TRACO 2017: Pancreatic cancer and Nanotechnology
Good. Afternoon everyone, welcome. To the final session of Traco 2017. I'm. Dr., Zia one of the faculty coordinators, of the course I would like to pass on some information, before we begin, the. Final exam will be posted, by December, 8th on the Traco, website, and. The exam can be taken until January, 12 2018. One. Question will be derived from each of the 26, lectures, if any. Of you have missed any of the lectures they will be posted on the NIH videocast, site for, your review. I believe, about 13 lectures have already been posted but, they shall all be posted. For. Your review, if, you, answer, 70%. Or better of the questions correct, you pass the exam and will be forwarded, a course, evaluation. Following. Successful completion of, both of those components a certificate. Of course completion. Will be mailed to you, in late January. In. Our first lecture is by dr. Purvis. Hussain, he. Is an investigator, in the center for cancer research pancreatic. Cancer unit laboratory. Of human, carcinogenesis. Unfortunately. He was not able to be here today so, we will play his videocast, from last year, his. Talk is entitled pancreatic. Cancer, current. Understanding. And future challenges, what I am going to do today is to give you a brief. Overview about, pancreatic. Cancer, what, are the current, progress, what, is our. Understanding. And what. Are the challenges, to. Improve. Survival, in this most. Lethal. Malignancy. In the world, so. Let me start with this. Slide which. Simply. Shows that, it's. The third. Leading. Cause of death. In. The, cause. Of cancer death in the United States you, see here it's fourth but then you combine both, men. And women it. Comes to the third leading cause of cancer. Deaths the, median survival, is less than six, months is, made fifty-three. Thousand new cases and, about, 41, thousand, deaths. Will occur in, 2016. And there. Is no effective, treatment so. So, everything. Seems. To be very disturbing. And. The. Most, disturbing is that according, to the estimate. It. Is going to be the second, leading. Cause of death due to cancer in the United States, by. 2030. So. What are the risk factors for, developing. Pancreatic. Cancer. These, are some of the risk factors for example smoking. Long-standing. Diabetes. Pancreatitis. Obesity. And also nan-oh, blood group oh and, these. Are some of the cancer. Syndrome, that are inherited, syndrome, where, you have mutations, in, in, different genes and that enhances, the risk of pancreatic, cancer for, example harry, did for a pancreatitis, where. You have mutation, in the trypsinogen. Gene. That enhances, the risk to, about. 54. So. In terms of treatment we, have not. Made. Much, advancement. In terms of treatment, if you look here, about. 20. Years ago, gemcitabine. Which, is a chemotherapeutic drug. Was, approved over. 5-fluorouracil. Because. It enhanced, the, survival, only by by. About one month and. Then. About, ten, years later our, law. Tunip which is a EGFR. Inhibitor. Was. Used in combination, of gemcitabine. Which. Enhance, the survival, only by. About. Two weeks and but. The situation, is so desperate desperate. That it was approved by. By. FDA and. It. It. Doesn't work in in. Advance cases, and, maybe. There is a subset, a small subset, that will respond. - these combinations. And then in 2011. A combination. Of different chemotherapeutic. Drug. And. The, combination, is called for fury knocks that, has the four different chemotherapeutic drugs. That responded, well, relatively, speaking in, pancreatic, cancer, as you can see here it enhanced. The survival, in advanced, cases. 11.1. Months as compared. To about six months by, gemcitabine, alone, but the problem, was, that it is highly, toxic for, majority, of the patients and then. Recently. In 2013. Albumin. Bound, nanoparticle. Paclitaxel. Was. Approved. In combination, of gemcitabine which. Enhanced, the. Survival, for few months as compared, to just, gemcitabine. But. It is less toxic, than fall fury Knox and then, most recently last. Year. Nano. Liposomal. Iron and he can and other two chemotherapeutic. Drugs. Were approved and. This. Also enhanced. The survival. By. About a. Couple. Of months when, when. Used in, combination, and. It has less, toxicity. So. As, we. Know that, the. Dismal, outcome, is because of the late diagnosis. If there is no early, detection marker. And I will show you one or two slides. About the situation. In. Terms of early detection and very, poor therapeutic, response as, I just mentioned, but. There are. About. 18, to 20% of the patients that, are, detected, early, and they, are detected early because they are.
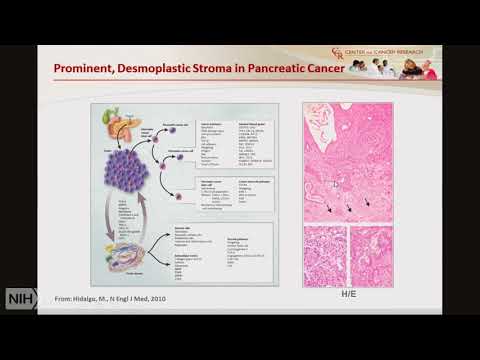
Examined. For something, else but, the physicians, finds, that okay you have the. Earliest stage of pancreatic. Cancer and these, early stage, pancreatic. Cancer, patients can. Qualify. For surgical. Resection so, you, will imagine that okay stage 1 it, is resected. Everything, is fine but. Still the median, survival even, in these 18 to 20% of the patients, is. Less than two years and. So. So majority of the patients about. 80%, of the patients show, recurrence, of the disease within. Two years, but. There is a small minority of patients. Resected. Patients, that do survive survive. For five years so, are there difference, among, these early stage, patients are there molecular. Differences, there. Are several, clinical. Prognostic. Factors, for example a stage, grade. Lymph, nor the spread and resection, margin, at the, time of resection, that, suggests. The, survival, good, or bad survival, in these cases however, we. Do see variable. Outcome sometimes, you see stage, one recession, margin, zero and the patient died within six months but in some cases the. Same stage same grade same, resection, margin estate as they survive for two years three years four, years of five years so, one of the hypothesis, is that there. Are molecular differences, in, tumors. That. Actually. Determines patient. Outcome. So. How, we can improve survival. In pancreatic cancer we. Know that you know we don't have. Early. Detection marker. Responses. Is, poor, so. We need, effective. Therapy, and for effective therapy, we need novel. Molecular, targets. That can be effectively, targeted, then. Molecular subtypes. That, molecular. Makeup, of the tumors, that will respond, to a specific drug, and, treatment. Selection can be done based, on. The molecular, subtypes. And then, drug. Delivery, so especially and, I will talk. A little bit more about the drug delivery and thank you already cancer it's very difficult, because. The. Tumor is, surrounded. By a huge, stroma. And it's, kind of highly protected, and it's also hypo, vascular, means it's not, vascularized. Well, so, when you give a treatment. The treatment, is not reaching. The tumor because. The the. Less number of blood. Vessels and also blood vessels are collapsed. So, there are some experiments, that have been done to. To. Reduce. Those, interstitial. Pressure on the, blood vessels and that has improved. Drug delivery, and also survival, which I will talk in a minute and also. We need early detection markers. The, biomarkers. That can in fact detect. Precancerous. Lesions, because. Once the, cancer is in stage, one or or, stage, 2 it's, already too late as I discussed. Earlier so, we need to have biomarkers. That can be a. That. Can identify. Patients. When they have precancerous, lesions, so for all these we. Need to. Understand, tumor, biology, that. Will involve, both genetics, epigenetics. Tumorous. Stromal interaction. Metabolic. Reprogramming. And transcriptional. Dis, regulations. So. Let's talk, about in. One, or two slides the biology, of pancreatic, cancer so. It is developed, by, well-defined. Precursor. Lesions, which. Are called pancreatic. Intraepithelial. Neoplasia. Collisions. This, is the normal. Are. Duct pancreatic. Duct here and this. Is the. Pancreatic. Intrepid failure neoplastic. Lesion, one. You, can see here some dysplastic, changes, going on this, is great - and this, is pen, in 3 which, is almost carcinoma. In situ and then, it. Penetrates. The basement membrane and it, forms, this. Glandular, like a structure, which, is called pancreatic. Ductal. Adenocarcinoma, and. Pedic. Or pancreatic, ductal adenocarcinoma is, the, most common, form of pancreatic cancer it's about more. Than 90 percent 90, to 95, percent and, there, are several. Well-defined. Genetic, alterations. That occurs, here, like telomere. Shortening, Kira's. Mutation. Is found. In about 90. To 95 percent of, the patients then. P16, alteration. Both die by. Epigenetic. Alterations. And mutations, and, also, p53. And s mark for mutation. That are found in about 50% of all, the pancreatic, cancer as. I. Mentioned this is just a a, section. Of the, pancreatic.
Ductal Adenocarcinoma which. Shows, huge. Stroma. These are the glands pancreatic, tumor, glands, and then, here. It shows that there. Is a strong. Interaction. Between tumor. And stromal, cells and they, support, each other so. As, I mentioned there is a lack of any. Marker. That, that. Can detect, this cancer, early, on there. Is only one, marker. That is clinically. Used, these. Days this, is carbohydrate. Antigen. 19-9. So. Anything. In the serum, if it is more than 37. Unit per ml, that, is a marker, of pancreatic. Cancer but, the problem, is that sensitivity, is only 80. Percent, and a, specificity, is also not, that good so this is not a very good marker, and it's. Also not just a specific for pancreatic, cancer, and also if you, compare, the malignant, versus, the benign pancreatic. Disease both. The sensitivity. And specificity. Is. Very. Low. In this cancer so. One. Study. That was published last year by ragu calories, lab what, they have found in the. Circulating, exosomes. If. They, are glycol, gleich, hand, positive, glycan is a proteoglycan. And they. Found that, majority. Of the pancreatic. Ductal adenocarcinoma. They. Have circulating. Exosomes. That have. That. Are, glycol. Positive. And this, is the discovery, cohort, and they validated, their findings, here this is healthy donor. Benign. Pancreatic. Disease this. Is this, is, precancerous. Pancreatic. Lesions and pancreatic. Ductal, adenocarcinoma and. As you can see here they, are showing 100 percent, specificity, and, 100. Percent sensitivity. So, if this is validated. Later. On in a larger. Cohort. Of pancreatic, ductal adenocarcinoma, then, this will be a very, good. Marker. For early. Detection. They. Also showed, that if. You have a. Lower, level. Of. Circulating. Exons, with. Which. Are glyphic and positive, then you have better survival, as compared. To the one that has, higher. Circulating. Exosome, that org like assume that, are clapping. And positive, but, they do not find that in the see a, 19-9. So, again they are saying that it is also a prognostic. Marker. In the pancreatic cancer, patients that. How. Well they are going to do I. Mentioned. Little bit about the. Molecular, makeup. That. Are there different, molecular subtypes. Of the, pancreatic, cancer, that, respond, differently to, treatment and they, have different, prognosis. And. These. Are some of the examples, so one, of the studies, by. Bert, vogelstein lab, at Johns. Hopkins what, they did they. Did. The genomic, analysis. Of only, 27. To. Mer sample. This. Study was done a, long time ago in, 2008. And what they found that each tumor, had. At least, 68. Or, 70. Different. Genetic, alterations, and these. All genetic, alteration. Responded. With 12 core. Signaling, pathway, that was, altered, but. At the same time what they also found that if, you compare two tumors. There. Were different, genes. That were ordered in each of these different pathways so. The, summary. Of this slide is that it's highly, heterogeneous. Tumor. And we. See a lot of heterogeneity. Inter. Tumor heterogeneity, in this cancer and, then, in 2011. Another. Study. Was published in which they they. Did. The gene expression, analysis, of pancreatic. Ductal, adenocarcinoma and. Based on the gene. Expression. They. Divided. These into. Three. Different subtypes. And one of the subtypes. Which. Is here quasi mesenchymal. Subtypes. Had the poorest, survival. Among. All these. Pancreatic. Cancer on, the. Same line, in. 2015. There. Was a, study. Where, they. They. Analyzed, the variation. In chromosomal, structure, and they. Identified. Four, different. Pancreatic. Ductal adenocarcinoma, subtypes. Stable. Scattered. Unstable. And local, based. On the on that degree of chromosomal, alterations and, the, unstable, type, was.
The One where the, most. Chromosomal. Variation. Occurred and this, was also associated, this. Subtype, was, with, the alterations. In that, DNA repair path and the. Patients which, represented. Unstable. Subtype. They, responded, very well to the Platinum, therapy. And they correlated, that because they have. Alterations. In that, DNA repair. Mechanism, that's why they, they. Responded, very well to that platinum, drug so that's how, these. Are providing, proof of concepts. That. Patients. For, example a stage 1 or stage 2 patients they. Should not be treated that they all are same patients, but, they they can be subgroup, based, on their genetic. Molecular. Makeup, and select. And stratify, those patients, that will respond, differently to, a specific. Drug. And. Then. Again. Last, year there. Was another, study. In which they they, virtually micro. Dissected, using, and. Computer. Algorithm. Where they, separated. The epithelial, tumor cells gene, expression, and the, stromal, gene, expression, and based, on the stromal. Gene expression, so they divided, in the into the activated. Stroma, and the in activator, and the normal stroma, and what they found that, no matter what the genetic makeup of the tumor is if the stromal. Tissue. Shows. That the, activated. Stromal. Gene. Expression, then, the survival, was poor, so what they were trying to say that, the studies where we take out the patient. Humor and do, the xenograft, studies, in the mice may. Not represent the actual. Tumor. Of the patients because if. The stromal. Gene. Expressions, are active, stroma, then, it will respond different, to. The treatment. And. Again. Recently, in 2016. So you can see this is a highly. Active, area, to, find out the, different subtypes, in in the pancreatic tumors. Tumors, so that they can improve, our. Treatment, so this is the the largest study, almost. 500. Patients. And, and. They, they, described, four, different subtypes, and they, were overlapping. With. The previous. Studies. Describing. Different, subtypes. And then. Based on the on the metabolism. And. And. The metabolic activations. This, was again divided. Into slope proliferating. Type glycolytic. And lipo jet and, this. Is the, last study on the, on the. Molecular. Subgroups. Of pancreatic. Cancer so, talking, about the. Difference, in metabolic. Programming. Or metabolic characteristics. Of, pancreatic, tumors, what. Studies. Have found that that. Pancreatic. Tumor. Cells they, reprogram. Their metabolic, pathways, and this, is a highly, complex slides but I would just. Just. Highlight, three points, and one. Is that you have increased, glycolysis, here. You. Also have see. The. Glycolysis. Intermediate, that goes into the non, oxidative. Segments. Of the. Phosphate. Pathway here. And, there. Is a alternate. Glutamine. Metabolism.
Here Which, is unique to the pancreatic, cancer where, glutamate. Is is converted. To aspartate. Catalyzed. By God - and this asked as part it is is shunted, out to the cytoplasm and, and. Gives, pyruvate. Through several sequential. Steps and. During. This process, NADPH. That is produced. Is used. For. The redox balance, instead, of the NADPH. That is used, by the pentose, phosphate. Pathway, here. So this is not active, here rather it has an alternate. Metabolism. So, these are the three points. Increased. Glycolysis. Or alternate. Glutamine, metabolism. And the, activation, of the non oxidative, pathways, these are the three important. Points and many. Of these enzymes, that. Are catalyzing. These, reactions, are, modulated. By chaos. So, if you remember about. 95%, of, the pancreatic. Tumors have, Kiera's mutations. And. Then. Recently. Last week. Know. In. August couple of months ago what. They found is, that as I mentioned that is true my is very, helpful. For, pancreatic. Tumors, and one of the important, components in the stroma, is the, pancreatic, is tailored cells and these, is tailored cells in, fact, produce alanine. And. It. Supports. Our. Cancer. Cells it fuels TCA, cycle supports. Lipid biosynthesis. And. Shunts glucose to serine and glycine. Biosynthesis. So, this is the. Latest, concept. In, in. In. Pancreatic, cancer. Metabolic. Rewiring where. The pancreatic. Still, its cells in the stroma, can. Support. The. And there is a positive, feedback loop, what. We don't know that. What makes tumor, cells, undergo. Autophagy, to. Produce. To. Activate. Pancreatic. Islet cells to, produce alanine. So. Let's talk a little bit more about stroma. Because, these are are some, of the of the latest progress, that have been made in pancreatic. Cancer, so, what has been found this is also a complex slide but I will make it highly. Simple. And, you. See here the environment, is immune, suppressive. So. Cd8. Positive T, cells is, inhibited. By T, reg cells MD SC and tumor associated, macrophages. That, is the one part, pancreatic. Is tailored cells which I just. Mention, that. Enhances. The. The. Tumor is stroma, which is called a small plastic reactions. And then, as I mentioned briefly that, due, to the highly. Decimal. Plastic stroma, you have accumulation. Of hyaluronic acid that. Increases. The interstitial. Fluid pressure. On the on the. Blood vessels and that. Inhibits. The drug flow, to the tumor. Okay. So. How we, can target a stroma. To. Enhance, drug delivery, so, there are two studies that have been done and this. Is the mouse. Model that have been used in this, study this mouse model has. Pancreas, a specific. Activation. Of Kara's and inactivation. Of p53 and they, develop pancreatic.
Ductal Adenocarcinoma which. Exactly. Recapitulates. The development, and progression of. Pancreatic. Cancer in human, so, they use these models. And what. They found is that if they inhibit, the. Stroma, through, inhibition, of Hedgehog, signaling. Pathway. Then. They. Increase the, survival. In this, mines with. Pancreatic cancer, and they, also decrease. The liver metastasis. Then. There was another study here. Where. They. They. Reduce the, hyaluronic acid. Deposition in, the stromal, by, using hyaluronidase. And, that. Reduced. The interstitial. Pressure and and. The, blood vessels that where we, are complexed. Got relaxed, and there, were more drug. Delivery. In the tumors and they found that, by doing this they enhance, survival, and they, also reduced, metastasis. So this this. Approach. Is currently, under phase 2 clinical trial, and they, have some some. Positive responses. But. There. Are two, other, studies, that, contradicts. These findings, by, saying that. Removing. A stroma or inhibiting. A stroma may not always be, beneficial. So in these two studies, that were published back-to-back, in cancer cells what. They shown using. A genetically, engineered mouse model, that, if you inhibit. Sonic. Hedgehog and reduce the stroma, in fact. The. Survival, goes, down. So, reduce. Survival. Here. And then, you also see increased. Metastasis. And you, see increase, the precancerous, lesions. Then. In another study, on the same line published. In the same journal back-to-back, they, showed the same thing that, my fibroblast. Depletion, enhanced, pancreatic. Ductal adenocarcinoma so. Morrow fibroblasts. Are the stromal, cells cells. In the dismal plastic stroma, and when. You inhibit. Those milo, fibroblasts, you, see increase, in, the. In the, tumorigenesis, and you, also see. Increase. Decreased. Survival, so. Based. On these. Contradictory. Studies, I think more. Work, needs to be done and more. To. Identify, that which patient, will respond. Better by, anti, stromal, therapy, so you have to reprogram, a stroma in order to, to. Enhance. Sensitivity. To, to, the drugs so to Morris normal interaction is a complex, and therapeutic, approaches. Targeting. A stroma needs caution, and may, require new, molecular, taxonomy in, pancreatic. Cancer. So. About one, of the glycoprotein. That. Is highly expressed on. Different. Tumor types and. This is mezzo, telling, this is a glycoprotein, that, was discovered, right here at NCI in Ida past-tense, lab. And, and. This was targeted. By using immunotoxin. In mesothelioma, which, very. Successfully. Which very good, results, so, so, this slide is just to show you that pancreatic. Cancer along with other cancers, also. Express. A large. Amount. Of mezzo talent so this is a good target, so, what so, Neil Hingorani and grooves did which. They. Engineered. T. Cells here. To. Target. Mesothelin. And they. Did this. And. This, worked very well by. Penetrating, through, the, very. Densest, Roma and also, immune suppressive, environment, and in fact the lies the tumor cells by binding to to, mesothelin, and also.
Also, Enhanced. Survival. In these mice so. This is also going to go in the clinical trial and let's, hope that that. Something. Comes. Up that. Will be beneficial to the patients. This. Is the. The. Highly. Advanced. Model. That. Has been developed recently just. In last one year which, is called organoid. Model and David. - Hasan, at Cold Spring Harbor Laboratory. Developed. This model and this is going to be the most. Important. Preclinical. Tool both. To understand, individual. Tumor biology and also. Identify. Drug. Targets, and test. Different novel. Targets, so, what happens that from. The patients, they, can take small part. Of the of the, tumors and they, can develop. In. Outside. In the, 3d, culture, exactly. The same. Architecture. Of tumour as you see in the patients and this, can be frozen and after. That if you transplant, that in, a mouse model ortho. Topically, it, starts, with that development, of precancerous, lesions, and going. To the to the advanced, cancer, so this is one, of the latest model, I just wanted to. Tell. You about. This how much time do we have Terry. Okay. Great so so I will take 10 more minutes and and. Please. Let me know if any, of these points what I'm trying. To make is not clear so I can stop, and then explain to, you. Inflammation. Is an important. Component. In pancreatic, cancer, development, and progression. Chronic. Pancreatitis as, I mentioned, enhances. The, risk of pancreatic cancer, there are several. Oncogenic. Pathways, that are constitutively. Active, for example NF Kappa B signaling. Desmo, plastic a stroma huge. Stroma, highly, active, inflammatory. Cells and. Inflammatory. Cytokines. Cox-2, and nos - these are the two. Enzymes. That catalyzes, different, reactions, that, produces, reactive, species. So. If. You, look at the cymatics. Of normal. Pancreatic. Interpret, allele neoplastic, lesions, precursor, lesions and. Fedak. You, see a stepwise, accumulation. Of inflammatory, changes here. Here. That, accumulates, or are, different. Stages.
There. Are also. Evidence. Which shows that as, I mentioned Kara's, mutation, is is, very important, it activates, Kira's, and more. Than 95 percent of the patients, have Kara's mutation, but. All the, mutated, Kara's, is not, homogeneous. So, what has been shown that mutated. Kara's, has, to reach a. Pathological. Level, of activation. And that. Is is provided. By, these reactive. Species that. Generated, during chronic, inflammation just. An example so, one cytokine. That. We, are working in my lab so I will give. You you. A. Brief. Overview with. Four or five slides that. What we are doing in our lab here at NCI, about. Pancreatic, cancer, and, we. Are interested, in one of the cytokines, that is called macrophage, migration, inhibitory. Factor. It's, a prole of inflammatory, cytokines. Expressed, in epithelial and inflammatory. Cells it. Was described, as a regulator, of immune response. Increased. Expression, in tumors. It. It also activates oncogenic. Signaling pathways. NF, kappa-b akt arc one two, enhances. Some. Of the enzymes, which I just mentioned it also, antagonize. P53, our b2f, pathways, and based. On these and other functions, it is described, as a link between inflammation. And cancer. So. We tested a simple hypothesis, myth. Contributes, to pancreatic cancer, progression. And predicts disease, outcome, these. Are the tumor and are just a non tumor tissue you see increased, expression of myth. In the tumors these are the normal pancreatic. Duct, when. You do the survival, analysis, this is the kaplan-meier, analysis. And if. You group. The patients, in two groups based on the myth, expression, level as high and low base. Using. Median value as the cutoff then. You see that the high myth, expressing, tumors. Patient. Have. Poorer, survival than, those patients, where, tumor expression, is is. Relatively. Lower and. This. Can be validated, in several, different, groups. Of patients or the cohorts and also the publicly available, datasets so. This simply, shows the Association, what, myth actually does so, we didn't in vivo experiments in which you, overexpress, myth, and Transplant, those cells in the in the tumors. You see that MIFF expression, tumors was, larger, and also. More metastasis. Here when. You look at the tumors. Histology they, are completely, different the. Control, tumors should. Highly. Differentiated. Or. A. Mod. Moderately. Differentiated or, well differentiated, but, the myth expressing, tumors, are poorly differentiated that, are more aggressive when. You look at the gene expression analysis. Between. These two tumors you, see, a. Change. In the global gene expression, profile and some of these genes genes. Represent, that. The. EMT, pathways, and me for expressing, tumors showed. Expression, of EMT, marker. Genes, then. In this paper. Recently. In nature cell biology, David, lied ins group showed that. In the pancreatic, cancer, patients, if the, circulating. Exosomes, have. Higher mics higher. Macrophage. Inhibitory. Factor, level then, they are more prone to metastasis. As I mentioned earlier. In my talk that. Pancreatic. Cancer patient earliest stage patients some, metastasized. Faster, some don't so, maybe we, beat, those patients, who, have Heim if expression, they are at increased risk of liver, metastasis. So. We wanted to see that what is the difference between. The tumors. Who. Are expressing, high level, and those, that are low level, so. We asked, two questions, what. Are the molecular distinctions, between these, two group of tumors and what, are the mechanistic, and functional, role of methane tumor progression. So. Highly, talented postdoc, in my lab did. The integrative. Transcriptomic, analysis, in which he analyzed these, two group of tumors by. MRNA. And micro RNA arrays and what. He. Tested the hypothesis, that MIFF regulates. Micro, RNA associated. With tumor progression and, disease aggressiveness, and he. Found several, differentially. Expressed micro. RNA between these two groups some, of them were also associated, with survival he validated these data in multiple cores, of pancreatic, ductal adenocarcinoma.
And. And what we found at the end that. Myth. Enhanced. A micro, RNA Mir 301, B and Mir, 301. B be. Targeted. One of the potential, tumor. Inhibitory, gene in our 3 C 2 which, has not been described, in pancreatic, cancer before and this, NR, 3 C 2 was. Inhibiting. EMT, so, if you have increase myth, it enhances. Mir 301, B and 301, B inhibits, and R 3 C 2 then there will be more EMT. And and, there will be more progression. And metastasis. So. The hypothesis. Is now that. Myth. In our, 3 C 2 signaling, is a potential, therapeutic target. In pancreatic cancer, so we went further and we, use the same model and we, deleted, the, myth, from, this kpc mouse model which i earlier described, and what. We found that, when you removed. Myth genetically. From these mice it, significantly. Enhanced. Survival, you can see and and, it also decreases. Metastasis. In these, mice and. Then. We, did some some. Molecular studies and you, found that myth. Deleted. Mice. They had increased, the akkadian. Decreased, zev 1 they, maintained an end catering and these are EMT. Marker, this is Western. Blot analysis, and then, again you. See reduced. Micro. RNA 301, B expression, in myth, diffusion, KPC mice and increase. The NR 3 c2 expression, just to to show that what we are showing, finding. In the, in the human tumors, the, same we. Are finding also in the disease model so. Now it's. A potential, target so. So, how we can go further the, further is that. Now. Use the myth specific, inhibitor, because you can delete gene in the patients so you have to use, these inhibitors, once the, mice developed. This tumor so that's what we are we, are doing there are several ways, you can use a small molecule meth, antagonist, which, we are using we, are also so. Using, anti, meth antibodies, which is in in clinical. Trial for or for other cancers and, in. Summary from. From. This last experiment. Which I showed you is, that a higher MIFF expression, is associated. With poorer outcome, MIFF. Enhances, growth and metastasis, of tumor. Xenografts, in mice may. Have driven signaling, inhibits, NR. 3 C 2 which is an anti. It's. A tumor suppressive, potential, tumor suppressive, genes by, up regulating, mir 301, B and. In. R 3 C 2 is a negative regulator, of EMT. Move, deficiency, increased, survival, and reduce, metastasis. Their. Form if Mir, 301, B in R 3 C 2 signaling, is, a potential, therapeutic target. So, I would like to end with this light which, I showed right in the beginning that. That in. Order to achieve early, detection and, effective. Therapy, we. Need to, understand. Extensively. Pancreatic. Tumor biology and in last. Six. To seven years there. Have been been. Enormous. Amount, of effort. That that. Are made in understanding. Pancreatic. Tumor biology and now, we, are at a point that we have several leads that. Can be be, taken into the preclinical, and and, clinical, studies so, I will stop here and take your. Question if you have any thank you very much for, your attention at 5:30. Yes. It does so. So. What happens that that. Makes. Enhances. The. The. The. Level of MD, SC and the. Level of t-regulatory cells in, the turrets so when we check the the tumors if. Expressing. And control. Tumors they, found, that when you remove meth you have less of those immune suppressive. Great. Thank you very much Thanks our next speaker is. Dr. marina. Dubrow. Of Ostia. She's. A senior, principal, investigator, and head of the immunology section. At the nanotechnology, characterization. Laboratory. Leidos, biomedical. In Frederick Maryland she. Leads a team of scientists, and technicians conducting. Preclinical. Studies to monitor adverse, effects, of nanoparticles. To the immune system both in vitro and in vivo, her, team strives to improve the understanding of mechanisms. Of nanoparticles. Nanoparticle. Immunotoxicity. She. Is a part of several nano, medicine working, groups and has published more than 45 peer-reviewed. Journal. Articles on this topic her. Talk is entitled, nanotechnology. For cancer therapy. Benefits. Concerns. And effects of the immune system and good, evening everyone. So. What I will talk today about the. Application.
Of Nanotechnology for. Cancer therapy I, will, try to give a very general. Overview of the field to, give you an idea what, nanotechnology, can do for cancer therapy, we'll talk about benefits. Of the technology and. Some. Concerns, and because my area, of expertise, is an immune. Immune. System, and immunotoxicity. All, of the examples that I will be using are from. The field of nanoparticle. Interaction, with the immune system this is the outline of my presentation and. For. Those who are watching us online I, will. Try to use. The. Laser. Pointer, here on the screen so you can. See this red dot moving so we'll talk about in a new technology, definitions, will talk about nanoparticles. And a daily, life I will give some examples, we'll talk about use of nanoparticles. And medical applications then, specifically, focus on cancer, diagnosis. And therapy, and. It. Review, the efforts on the immune, system so what is nanotechnology and. What are the nanoparticles. There is no. Universal. Definition one. Of the very commonly used definition. Is from the National nanotechnology initiative. They. Defines, nanoparticles. As objects. With a size and at least one dimension between, 1 and 100 nanometers. But, as you will, see later from my presentation. Many, of the nanoparticle. Formulations. That are approved, for the clinical, use are a little bit larger, so they were or average, size of. These. Materials, that are currently used in the clinic to treat cancer. And then also other. Diseases. Is. 350. Nanometers and, below. And for this reason, United. States Food and Drug Administration came. Up with a different, definition where. They say that they, will consider a material. With. The size up to 1 micrometer, if, that, size brings, the size related, protégée which is a main at the property. Of a nanotechnology. So. There are lot of ana particles, in daily life here you see, some. Of the products, that are used on. The market, they. Are manufactured. By more, than 800, companies. In, many, countries around the world, include. Clothing, would, wound. Racing, machine liners. Sunglasses. Sporting. Equipment. You. May go online in, Google and you, may find a lot of the silver nanoparticles for, example, or gold nanoparticles. In this health. Stores, online that. Sell. Them for improving, mental health, and, the immunity, and everything, else. But. Then we develop these materials for clinical applications, we have to go through very rigorous, characterization. Of efficacy and the safety so, these are other examples of, the nanoparticles in daily life they include, metal. Oxides such as titanium. And zinc oxide in. Sunscreens. So you differentiate. Within the nanotechnology-based. Sunscreens. And the regular sunscreen by, the color if you apply sunscreen on the skin and you see why it marks it's not, mana and if you apply sunscreen on the skin and there is no white marks, then they contain, these nano sized materials. Interestingly. A. Lot of the cosmetics. Companies. Like L'Oreal include.
Nanoparticles. In the form of these lipids ohms and amongst the, lipid based physicals. In their creams because these materials improve. The penetration, of, whatever. Is in this lipids, and in, the skin our. Carbon, nanotubes, on, the other hand are very durable, and they are used as a structural, materials, to my knowledge there is no carbon. Nanotube, based, nano, medicine. Approved. For the clinical, use most, of these materials are used in industrial applications, however. In basic, research if, you search PubMed, you will find a lot of the applications were, even, nanotubes, and graphene. Particles. Are used for delivery, of the drugs for. Cancer therapy. Here. I would like to give you some idea of the distribution of, the nanoparticles, in, current, drug products, for, this I use this paper recently. Published in Nature nanotechnology. By, Catherine Tyler and her, colleagues if you look in the top row of this pie chart then. You will see the wood. Is what. Type of the nanomaterials. Are used for drug delivery and, you see this large piece. This. Separated. By the. You. Know by, the years you, will see that majority, of these applications. Are Lepus, ohms, followed. By nano, crystals, followed, by emulsions. And iron. Polymer. Complexes, and other, types of the materials, if, we look at the indication. The majority, of indication. Is cancer, followed, by inflammatory. Diseases. Followed, by. Infections. And anaemia, treatment, and, finally, if we look at the route of administration that. Majority of the material. Than. Our materials, that I used in drug products, I intended. For systemic administration their. Intravenous. Route the, next second. Popular route is, oral. Route. Nanoparticles. Are used in, cancer. Therapy all you. Know the main reason in, even for, the use of nanoparticles. And other types of therapies, is because. Nanomaterials. Can improve, cellulite of hydrophobic, drugs a lot of the small molecule, drugs are insoluble. And then we use them for basic, research we can use DMSO, to solubilize but. We cannot, inject. DMSO, intubations, nanoparticles. Can help so this, problem. And a burkha overcome, other, barriers. They, also over, this multifunctional. Capability. This is a schematic, example. From dr., McNeil's review, that shows you the multifunctional. Nanoparticle. Where basically, we have a core, particle, and then it has a drugs attached to the particle, has polyethylene. Glycol, on the serve it has an image in agent and a target in moiety, so there are variety of. Active. Pharmaceutical ingredients. Which in theory, one can put on the surface of the same particle, and could. Deliver these, materials, into, the target cells. Nanoparticles. Are used for a boutique tasks, such as incensing, computation, triggered. Responses. And in. The field of cancer therapy tumor. Targeting, is one. Of the attraction. Of, reasons, for using nanotechnology. One. Of the benefits, of using. Nanoparticles is, the reduced toxicity, and on this slide I will show you two example, one from the small molecule, drug, and another, one from. The drugs which belong to the category of, therapeutic, proteins, and what I do I will compare traditional, formulation, of this drug with, a nanotechnology. Formulation, so, small molecule, in a whole of you are familiar with, this drug, doxorubicin. Which is used for cancer therapy the. Nanotechnology. Formulation. Of this drug is known as Dogzilla, pegylated liposomal, doxorubicin. He, we have doxorubicin. Inside, of the. Lepus. Ohm that also has polyethylene. Glycol, on the surface, to improve circulation time. And thus toughness or office, particle. So, the d IC, and pcs, stand for disseminated, intravascular coagulation. And, procoagulant. Activity. Respectively. So these two, types, of the immunotoxicity. Are, also, known as consumptive. Kalila pathway if. Drug. Or some cancer, cells may induce di c. That.
Induces. Leukocytes, to express this phospholipid, protein. Complex, on the surface that serves. As a platform for, assembly. Of the blood, coagulation, factors, the trigger blood coagulation, and because, of this massive, coagulation. Later. On in the course of this toxicity. Patients. Are dying from bleeding because of the massive consumption of coagulation, factors, so basically these types of toxicity changes. The, hemostasis. In. The, in. The blood so this type of toxicity, is very common for. Doxorubicin. In fact many, especially, in the acute, myeloid, leukemia, patients, and the blood cancers, and this drug is used. The. Patients, often. Have to be. Withdrawn from this drug, because of the induction. Of the GIC known. Of such reports. Are. Available. With liposomal. Formulation. Of doxorubicin. In the, area of therapeutic, protein, I would, like to use example, of the tumor necrosis factor-alpha. It's a cytokine, it's a recombinant, protein, that, was tested in clinical trials. In. 80's. And it. Failed because. Of the systemic, inflammatory, response. However. There, is a small biotechnology. Company, here. In, Maryland. Site. Immune Sciences so what's item incises, team did they, put tumor, necrosis factor on. The surface of the pegylated colloidal, gold, and now this concept successfully. Passed phase 1. Clinical trials. Without affecting. Without inducing, systemic. Inflammation, systemic. Inflammatory responses. Very. Popular. Area in the cancer therapeutics. Days is, immunotherapy. Right we are here in this fascinating. Reports, about success, of the, Emunah therapies, but. Nanoparticles. Have a. Lot. Of benefit some particles, may be used for specific delivery, of the muna therapeutics, but in this, case. I would like to share with you some of this hidden, and yet unexplained. Unknown, mechanistically. Effects, from the nanoparticle. So remember I told you about dogs so it's a regulated. The personal doxorubicin. If. The study is conducted in, a thymic, mice then. In. This case they use CT 26. Colon. Carcinoma, model this is a study recently published, by salami, at immune and you, see there is no difference between the. Doxorubicin. And dog Co at those. Of 5 milligram, per kilogram and, even untreated, mice so the tumors continue to grow however. If they conduct, the same study in the same cancer model, and use the same doses. Of the drug in, the. Immunocompetent. Animals, and about sea animals then. You see that, in, the dog cell treatment, they absorb, delay, in the tumor growth so eventually tumors, grow but there. Is a delay, in the tumor growth more. Others so what they did they now take dogs'll. And they combine, it with known. Immunotherapy. You --tx with anti PD one and a CT LFO, and ox40. Ligand, and you see them in the combination, with, ntpd, one they, out. Of 12 animals, 11, achieved. Complete response, so this is tremendous, improvement, in this model. Of colorectal cancer the. Mechanism. Of this improvement is not understood, but there is increasing number, of the studies that, show. Dog. Seal that show Abraxane, another poster, child of cancer, nana medicine. For. Therefore the abilities, to improve. Outcomes. Of the, known immunotherapies. And in fact what you see. Here is some example. Of of. The phase 1 clinical trial. Which investigates, a Brooks, in, combination, with MVPD one for, metastatic. Breast cancer, again, the science has to, be. Still, established, and the mechanisms, of this potentiation, have to be understood, but this is very interesting. Very exciting. Observation. Nanoparticles. Are. Increasingly, used, for delivery, of the vaccines. Particles. With a small, size. By. Small I mean the, size less than 100, nanometers, after, subcutaneous. So intradermal. Administration, show, active. Trafficking, into the lymph node and lymph, nodes into the lymphatic and lymphatic, drainage offers. Additional, benefit. For the vaccines so, here I used the, example. Some. Of the data, that was recently, published. In this case it was systemically, administered. Republic's, that carried humour specific, RNA and. The. Study showed. That. Lippo, plaques protected, RNA from degradation so, RNA a device is, unstable, and, the. This. Particles, delivered, RNA, into. Antigen, presenting cells, and. It led to the greater production, of interferon, responses, and induction of the stronger, effector, and memory t-cells. I, talked, about the. Lymphatic. Delivery briefly, so, here I would like to use example, of the study where, the. Nanomaterials. Were the size of 25, and 100, nanometers were used and these particles, were labeled, with a fluorescent tag. And so you see that after injection, into the tail when the, I, mean. The I apologize. No not a little well in the tail in the subcutaneous space, of the tail you see that 100, nanometer, particles. Stayed at the site of injection and, the particles, were the size 25, nanometers are, traveled from the lymphatic and then were detected, in the lymph nodes. However. Very, often the, benefits, of Nana technology.
Are Also. Come. Along. With the concerns, about the safety of these materials, and here I would like to show you some of the studies showing, how toxicity. Can. Relocate. Okay change depending. On the type of the nanotechnology, carriers. For the same drug again, I'm using the example, of this small, molecular drug doxorubicin. It's very well studied there are a lot of data, in the literature about, using, this material on different types, of nanotechnology. Platforms, then, this drug is used as a small molecule drug, it accumulates, in the heart tissue that emulates in the bone marrow and as. A result, the. Mindless separation, and Gardea toxicity, are the main those limiting, toxicity for this material. Regulated. Liposomal doxorubicin. Remember I told you overcome, that. GIC, type. Of toxicity, however, this. Formulation. Now, results. In that, the drug accumulates. In the skin so, that cardio, toxicity and mild a separation, are decreased. Especially, cardio toxicity is decreased which is a big help for the cancer patients however it. Comes with this inflammatory. Palmer. Plant illiteracy, stage, also, known as a hand. And foot syndrome it's very painful and, patients. Have to be on the immunosuppressive. Therapy, to improve the quality of their life so, the. Cardio, toxicity is, a greater concern, and, it was overcome, by this formulation, however, additional, concern, was created, by the formulation. Of this drug and a lot of researchers worldwide, are. Currently, working on. Changing. Or optimizing, this formulation, so, that, this toxicity, is a still not. The problem but so that PPE. Syndrome. Is also. Solved. Same. Drug. Formulated. Using sonic related, nanoparticles. Now was accumulated in, the kidney so, all of this is example then all ofus and desirable, toxicities, were resolved, however the, nephrotoxicity. Was, the result so, the drug can relocate alone, with the particles, and particles, may change by distribution, of the drug and we. Need, to, realize it we need to appreciate and understand, that then we use variety. Of the nanotechnology carriers. For, the, drug, delivery now. Let's. Shift. To, the interaction. Between nanoparticles. And the immune system, it's. A very big area and what. I would like to do I would like to focus on several, aspects. So I chose the plasma, proteins, because I mentioned, to you about, the particles, ability, to change drug by distribution, so how this happens we'll talk about the plasma proteins and how they affect by distribution, of the nanoparticles, and they uptake, by manna nuclear phagocytic system we'll talk about particle, attacks on erythrocytes. Interaction. With the blood coagulation, system and I chose various, components, included light it's leukocytes, and the filial cells will, talk about allergic, reactions, to nanomaterials, and. Cytokine. Responses, and the immunogenicity. So. Proteins. You know the plasma, proteome contains, what around roughly. About 3,000. Different, types of the proteins and albumins. And goblins are the dominant, proteins, but there are a lot of the other proteins, in our plasma, which may change during. Physiological. Cycles, which may change due. To the level of stress the diet that people have the genetic, diversity and so forth and this is very important, because nanoparticles. If unless. The surface, is protected from with. A hydrophilic, polymers. Nanoparticles. Bind these proteins and there is a bi-directional, interaction. Between the particles and the proteins, so, particles, may interact with coagulation, factors. They. May activate. Or inhibit coagulation. They, may bind. Complement. Proteins and, result and complement, activation, they. May.
Change. The conformation. Of the proteins, that will, affect, the stability of the protein the conformation, of the protein its function, on, the other hand proteins. Also. Have, some effects on the particles, and that the particle, size may change that. By distribution, will, change the zeta potential, the. Let. May interfere with a targeting. Of the particles, and so forth so, here I show you example. From the book chapter wrote by. Tommy. Said, er pal where, they use the modeling, to show. On the particle, size so, they choose the 5 10 25, and 50 nanometer, particle, how, the two. Commonly. Present. Proteins. In the plasma would. Behave, and, serum. Albumin is, shown. In. Red so, you see that as a particle, size increases there, are more buildings that can bind, to. The surface. Of this particle, this. Is example, of. The. Protein. Effect, on the particle, physical chemical properties, from the study, that my team conducted, several, years, ago in this case we use 13, nanometer. Colloidal, gold, nanoparticles. And. Before. We, incubation. With plasma will, do the hydrogen, we measure the hydrodynamics size, by the Nemec light scattering, and we see that, the size is 33. Nanometers which, is very close to their nominal size right, but after incubation with, plasma, particle, size changes, to 76, millimeters, that's, because all of these proteins, that will absorb absorbed. On the particle, surface increase, form. This protein corona and increase the hydrodynamic size, of the particle, so the reason why it is important, is because our, body will not see through t nanometer, gold our body will see 76. And a meter particle and, in, the places, where the size is important. For. Passing. Through the. Healthiness. Tration, of. In, the. Tumor. Vasculature, the. Difference, between 33, and 76. Centimeters, may be substantial. Protein. Binding, effects. By distribution. Here, I show you example, of the study that we conducted in. Collaboration, with site immune Sciences, society, mune product, is a colloidal, gold, coated, with polyethylene, glycol, and, this. Is a particle, which. Is a counterpart. Same. A size same, composition, except, that it is missing, a polyethylene, glycol. Coating, you, see that, if we. Look. At the protein. Binding and in this case we're using two dimensional, references, each of this black sport corresponds. To the certain type of the blood of a protein. Bound, to the particle, surface there are more proteins, that I detected, on the surface of the uncoated particle. That, we. Can, see on the surface of pegylated particle, so Pig elation does not completely, prevent protein binding but it significantly. Reduced the amount of protein bound to the particles. Now, we look, at the uptake, of these particles, by macrophages, while, uncoated, particle. Are readily taken up by macrophages the pegylated particles. Do not and then, if these particles, are injected, in liver you see the color of the liver and the spleen in animals. That received, just a colloidal gold, the reason why this color is black is because gold. Normally. It has a pink color but, then, it is taken up by macrophages, it, aggregates, and the aggregation. Changes, size, obviously and changes color so this is why this. Black color basically, means that the liver and spleen are, loaded. With the gold nanoparticles, however. Then pegylated, version, of the same particle, is injected. Then, they stay in circulation longer. So eventually peg, does not completely prevent uptake. By the man a nuclear phagocytic, system, but it delays and so while this particle, stay in circulation longer. It allows them. Greater. Time, to, be distributed. To the tumor. Tissue. And. Deliver, the drug there. There. Are different models of the nanoparticles uptake. By the mononuclear, phagocytic, system, William, Zamboni from the University, of North Carolina. Suggested. These two models one, is a capture, and the other one is hijacking. So in the capture model particle. Is flowing. In the bloodstream and is. Captured, by. MPs. Like tumor resident, macrophages, or Cooper. Cells in the liver or. Resident. Macrophages, in them in the spleen in the hijacking. Model, particle. Is captured, by the monocytes. In the bloodstream and then it is the monocyte. That delivers, this particle, to the humoral, liver ozpin, right, so you understood the difference, so either particle, is go into the tissue and then is captured, by immune. Cells there or, immune, cells do their job right there in the bloodstream and then it's the immune cell that carries it to the tissue it's very interesting concept and actually this hijacking. Model. Is very, actively. Being. Explored currently, for, the drug delivery, especially the drug delivery to the to, the brain for, the. Some. Other vacation, were immune cells, are present. In the large quantity, now.
Let's Talk about some, toxicities. And undesirable, interaction. Between particles and, the immune cells a malicious, hemolysis, is a type of toxicity, that, has a variety of the. Symptoms. In the patients of it occurs it may be fatal depending, on the degree degree, of the hemolysis and it refers to the damage of red blood cells interaction. Between nanoparticles. And red blood cells depends, on the particle, size and also it depends on the particle, surface charge, in that, the. Cationic. Particles, or hemolytic. Well they neutral, and anionic, counterparts. Are not and. Here. I would like to show you a case study using. Pam. Dendrimers, we. Used palm dendrimers, with various hydrodynamics, eyes, the GE refers to the generation, the greater the gene number the larger, the particle, is and then we used three, different types of the surface coatings, and so what we observe is, the small, particles, did not demonstrate resides, but the. Large, particles, did and then if we look at the surface charge then, I mean terminated. The cationic, particles, damaged there it resides while they're in. A unique, and neutral counterparts. Did, not. Calculation. System, as I mentioned. All this exists, in the balanced. State between Pro and anticoagulant. Activity. And. Some. Particles, may shift, this, balance. Either towards, pro or, anticoagulant. Again in the, state of the disease, right, if there, is a procoagulant. If, the patient isn't procoagulant, state, like a majority. Of the. Patience. Then, the nanoparticle, with anticoagulant, property, would be beneficial however. If. There is a healthy, individual, in which there is a good balance between Pro and anti garland activity, and particle, has andraka, gland activity it may lead to the bleeding right so it's always about the balance, and the shift in the balance what, we know occurrence, from, variety, of studies, that. Particles. May have. Various. Types of effects, on different. Players of the coagulation system. Particles. May interact with coagulation, factor, and result in contact activation they. May bind and deplete coagulation, factor, which results, in bleeding they, may be toxic, - and ethereal cells they may induce inflammation. And, oxidative stress, and intestinal, cells and they also can affect applied, leads either by direct activation, or indirect activation. Here. I would like to share with you some examples of the studies that my team conducted, again, we are using the and rumors you. See that the small particles. Do not induce platelet, aggregation while. Their larger, counterparts, do. It as well as sa positive control, and this is the scanning. Electron microscopy. I'm, showing. The particles. I'm sorry platelets, in the control. Samples, and after, treatment with the dendrimer so you see that the flightless basically formed the slight clamps. Now if we look at the ethics, of the terminal, groups we use here generation. 6 particles. So all of them have the same hydrodynamics, eyes, they, are only different by the type of terminal. Groups we, see that in neutral, and anionic. Particles, do not induce. Platelet, aggregation but, they are cationic, counterparts. Do and. This. Study was conducted in. Collaboration with, dr. Daniel Patrick who, at the time worked in at, the NCL so, what we what. He did he, used this. Cationic. Dendrimers. And we. Might imitate. Platelet. Aggregation. Now, then, they masked, 25, percent, of amines on these dendrimers, we, noticed, some change in the calculation, as else they kept masks, and, the. Surface. Amines the toxicity, decreased. So the zeta potential is very important, the last surface amines are available, the last platelet, aggregation this, is very important, for the drug delivery because, it.
Is Chemically, you, cannot conjugate. Drug. To, carboxyl. Group. Or two, hydroxyl. Groups but, amines, are very good. Surface. Monitors, for attachment, for the attachment. Of the drugs and targeting. Ligands and if. There are unreacted. Amines when the particle, surface they may result, in toxicity so the basically the take-home message, from this status, if cationic. Particles, are used for drug delivery after, reaction. With all of the targeting monitors with all of the drugs any, uncovered. A means should be inactivated. To reduce the ability of this particle, to interact with the Ritter sites to interact with the platelets to avoid hemolysis, and tram. Bhutanese idiom, dendrimers. Come in different sizes, but also in different. Chemical. Composition. So here I show you comparison. Between. Transcendental. Merson tammim dendrimers, we. Compare the, particles, of. Same. Size and. What. We see that for both types, of the materials, the larger, the particle, the, greater the, reactivity. With platelets. However. Now if within the same size of the particle, if we, look at a. Comparison. When the tree resin and param dendrimers, then we see that pam dendrimers, are more reactive and, if you look here at the schematic. Of these materials, you see that at the same generation. Simply, because the starting, material. Is different. We, have less, number of a surface amines, on the tree resin dendrimers, and and Thummim dendrimers and this is most likely the reason for, why the same size of the particle, the same generation, and the same surface properties, but has little, different, biological, outcome. This study was conducted in, collaboration with, dr. Eric Simon again at, Texas, Christian, University. So, now let's talk. About the leucocyte procoagulant activity. That's in other. Factor. That contributes. To the blood calculation. A lot of the cells that includes leukocytes, that also include cancer cells in. The resident, leukocytes, do not express, pro-government. Activity, complex on the surface, but, after activation, with some, inflammation. Inducing. Ligand such as bacteria live a polysaccharide, for, example, or in case of the cancer cells after treatment, with cytotoxic. Drugs such, as doxorubicin. This, cells made for expressed so-called procoagulant activity. Complex, which is composed. Of the protein tissue factor in phosphatidyl. Serine. And this. Complex. Serves as a platform for, activation, of the, blood coagulation, factors, again, as a model, I am using of. Dendrimers. And as, you see as the particle size. Increases. The ability of this particle, to induce procoagulant. Activity. Increases. And if we look at the effects, of the surface charge then, we, see the same types of the effects that we, observed with hemolysis, and platelet aggregation right, for the particles, of the same generation only I mean terminated, particles, induces, toxicity, but the, hydroxy. And carboxyl, terminated. Counterparts, do not. Nanoparticles. May have, undesirable.
Effects On and athelia cells they may induce, inflammatory. Response, or they may have some direct toxicity on the ethereal cells, and depending. On the type of the activation. Of the endothelial cells it, may lead to the endothelial cell, dysfunction. Cells may die and that will result. Undesirable. Side, effect. Her. Nanoparticle. Allergenicity. Is another. Complex. Area. That, is being actively, investigated. What. I would like to the. Message that I would like to deliver in this presentation, is that particles. Can be engineered to. Inhibit allergic, responses, there are a lot of tile orogenic, particles, or particles, carry. The. Drugs. That interfere with. Allergic. Responses. However, some particles, may exaggerate. Allergy. To traditional, allergens. And may. Also be. Under chaining. This table, is from. The recent publication by Professor, Africa, Gonzalez, Fernandez, and you. See there are different types of at a classical, allergic. Reactions, a type 1 immediate, hypersensitivity type. 2 set at oxide hypersensitivity. Type 3 immune, complex mediated, hypersensitivity. And type 4 delayed, type hypersensitivity. And, there is this pseudo allergy, so the pseudo allergy, is. A. Type. Of toxicity. That does. Not involve. T-cells. So immunoglobulins. Specific. To allergen, but it is triggered by the activation, of the complement, in other terminology. For this type of reaction is carpel complement, activation related. To the allergen, when. It comes to nanomaterials. Carpa. Is the. Best studied. Response. To, nanoparticles and. There are also limited. Number of reports, about the type. 4 delay type hypersensitivity, and. I will show you some of the examples I, am not aware of the type 1 type 2 and type 3. Allergic. Responses, to nanomaterials, however, in the area of environmental and. Occupational. Toxicology. We know that some of the nanoparticles like, carbon black particles, that are present in the exhaust in the in the air may, exaggerate, the, asthma. And allergic, responses. Of the already sensitized. Individuals. So. This is example. Of the. Response. To. Pegylated, liposomal, doxorubicin. This, is a study was gonna call study was conducted in, patients, what, you see the numbers over here refer, to different. Patients, and white. Bars, show, the baseline. Levels, of the complement, terminal, complex in the plasma of these patients. The, black bars show, the levels. Of FISC complement, terminal, complex ten, minutes after, ministration, of the pg-rated liposomal, doxorubicin. And you see that response varies, between the donors but many, donors. Who, developed, a. Type. Like. Anaphylaxis, type, reactions, also had high levels of the complement, split product, and this is a type of toxicity, is dose, limiting toxicity for. Pegylated, liposomal, doxorubicin. Now, one, thing that we know about complement. That in addition to this undesirable, hydron. Sensitivity, reactions, methyl axis complement. Is also, very important, for building. Humoral. And cellular, immune response, therefore. For. Nanotechnology.
What. We, are doing. If, we know that. Particles. Are, intended. For systemic, administration. And, non vaccine, on immunotherapy. Type of reaction, then, enough, alaric activation, of complement should be avoided, to avoid unethical, axis and hypersensitivity. Reaction, however if, particles, activate. Complement, if we change their route of administration to. Subcutaneous. Intradermal. They may actually benefit, vaccine efficacy and, there are examples, in, the, literature. Where. Particles. Which, are able to activate the complement are successfully, used in vaccines. This, is an example, of the study that shows delayed, type, hypersensitivity. To, nanomaterials. To. My knowledge it's only one. Case. Of this necrotizing, dermatitis. That, was. Described. In the literature in. Response, to the end rumors and was actually, very interesting study, it was conducted in Japan the, student. Reported. To. Emergency. Room with, fever chills, and. Erythema. And this fused, bula. On his skin they. Could not understand, the mechanism, but every time this, person. Reported. Back to the lab he, experienced, the same type of reaction and this, lab was processing, large quantity. Of the dendrimers, it was a cyst you know systemic, lab no other people in the lab had the same reaction, and, worldwide. A lot of people are using with dendrimers, a large quantities, of these materials, have been produced, so, still this is very interesting clinical report, without well. Understood. Mechanism. Behind it however the, message that we learn from, this, diet is first of all somebody. Has to do more, investigation. To understand, this type of toxicity, and 2 then we work with nanomaterials. It's very important, to use personal protective equipment and, all of the other. Measures. To prevent, exposure. Undesirable. Inhalation, of steel and skin absorption of these materials, now, let's talk about the induction of the cytokines, before, we talk about nanomaterials. I would like to use this example from. Biotechnology, products, there was a super monoclonal antibody. TGN, 1412, in Europe it's a antibody. That specifically. Recognized, the cd28. On the surface of the T cells preclinical. Studies were conducted in non-human. Primates, and in rodents and did not reveal cytokine, storm however. Then, this, material, then this antibody. Was used in the clinical trials, all. Healthy. Volunteers. Who participated, in the study in the study developed. This. Necrosis. Due to the overwhelming cytokine. Production and, specifically, the overwhelming. Levels of the tumor necrosis factor-alpha. That. Result, in multiple organ, failure many, of this. Patients. Spend. Months. In the intensive care units. And it. Took several years to, understand, the, mechanism. For this toxicity turned. Out that. This. Antibody. Induced, high level of cytokines, and specifically, tumor necrosis factor alpha, and, this type of toxicity, is easily. Identified, using human. Peripheral, blood mononuclear cells. N
2017-12-07 14:45



As much as I am grateful that these lectures are shared they also remind me of how slow moving, opaque and unable to stimulate or excite the mind biological research is, especially when it comes to marrying insights from molecular dynamics/ab inito simulations, genomic data/informatics, with these incredibly broad and macroscopic views of the data. Also another thing that I have noticed is that almost rarely does anyone give some thought to the chemical/molecular structure of drugs and other compounds and most research is literally just a more dressed up "spray n pray" approach with no appreciation for the context of things. All that complexity just reduced to a bar graph. There must be a better way to not only get that kind of insight but to also capture and distill the most meaningful of dynamics at multiple structural scales and resolutions with better tools.