Howard Chang (Stanford, HHMI) 3: LncRNA Function at the DNA Level: PVT1
I, am. Howard Chang I'm a professor, at Stanford University. And investigator, with a Howard Hughes Medical Institute. This. Is part three of my AI biology, talks and this talk will focus on sis. Action, of long. Non-coding RNA, genes. The. World RNAs, been expanding, in an amazing. Rate over the last decade, it's, not believed that, the human genome encodes nearly, 60-thousand, long, non-coding RNA, genes, this. Picture, shows some of the history of this particular field and we start with this discover, a singular, RNA genes back in the 80s and 90s to, the present. Day explosion. Some. Of the sort of better worked out examples, are shown and, they're, principally. Working on chromatin, the guys by no means their only mode of action. Our. Work has focused on try to understand the mechanisms, of a select number of linked, rnas of biological, significance and. Also developing technologies, so, that other investigators can, tackle their favorite, long non-coding RNAs, a very. Important, sort of idea. Is that. Each of these linked RNA genes, has. Its, own set of regulatory elements that, are needed to for the production, of the long non-coding RNA, so. That means that each linked. RNA, would. Have its own promoter, its. Own enhancers. Splicing. Sites and other, features to make sure that this RNA, gets made and process. And sometimes. Is Ashley. These regulatory, elements that provide the functionality of, the, long non-coding RNA, locus not, necessarily, the RNA product. Some. Examples. From many investigators, our charters showed these, kinds of examples and today. We're, going to focus on a very salient example, and. That isn't a link Arnie and I'll tell you about the story of how we came to that discovery. So. Faced, with this explosion, of long non-coding RNA, genes a few, years ago we set out to determined, which, link arms were actually important, for cell growth and function, we. Teamed up with colleagues, at UCSF. Developed, a. Spirt interference. Technology. For, targeting link rnase this. Particular method uses. A, repurposes. A dead past nine from the CRISPR caste system as a DNA, targeting platform, we. Can fuse that dead caste nine to, a crap, silencer, domain and now, we recruit, a silencing.
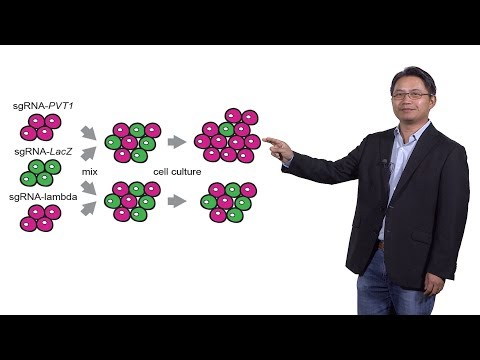
Complex, To. Wherever we targeted, this is the kind of epigenome. Editing. And this is called CRISPR interference. And. So wherever we target, this. Fusion, construct, we basically blocked a transcription, of the link RNA and gene basically. And we can then look at the consequence, of the. Loss of this link RNA transcription event we. Design us the. Guide. RNA is targeting. 16,000. Plus, female, link rnase particularly, those that may have a few effect on cancer and we've had ten guys each and so, this large this, screen was then conducted in. A seven, different cell types and, we got a quite striking result first, is that we identified, nearly 500. Essential. So essential, link RNA low sigh daddies, means that in the if we target, that link RNA the cells would start doing less well they would basically drop out from the population, and so there's something important, going on for this low sign the. Second point is that these linked RNA, essential. Hits we're actually oftentimes, present. In one cell type only and this, is a kind of a surprising, result because, for, the equivalent, screen, in protein. Coding genes these. Sol essential genes would be things, are involved in cell cycle progression the. Proteosome, really, important housekeeping, events and they'll be in common, across, multiple, cell types but in contrast these linked RNA hits are actually, in one cell type only this, will be the example where in one cell type a if, he signs as linked RNA gene doping. With. A cell growth defect, and also multiple, gene, expression, changes made, in the second cell type basically, it's not required no, growth defect, no gene expression changes and so there's a unique, level of South high specificity, that's, revealed in this kind of functional screen. In. Addition to genes. That, basically. Are important for cell growth that is the cell growth with slowdown in their absence we. Also got hits of the reverse kind and the, strongest, hit that. Enhance. Outgrowth entire, in the entire genome, why scream was, this particular link, RNA called PvP, one you. Can see some primary data here this is in the beginning of the experiment we transduce. The entire library, over time, we'll see what happens the cells that are missing PP t1 and you can see that basically, their, relative, in Richmond increases. In the population in, these two kinds of breast in this breast cancer cell and so are other cell types but, not in some additional cell types okay so the absence, of this RNA seems to make the. Cells, grow better, this. Is a very salient finding, because PV, t1, is perhaps the, first link. RNA gene, implicated. By human cancer genetics. DVT, one is. Ashley, its locus, is a place. It's resides. At, a translocation. Breakpoint. For a kind of human cancer called, Burkett's lymphoma. And. It's right next to there very important oncogene called Mik 53. Kilobases, downstream. And, in fact this translocation. Actually. Led to the cloning, of Mik and actually, wanted the identification, to make us a human oncogene almost. Thirty years ago so they're very important, sort, of landmark, in cancer, genetics, it's. Knowing that this. Locus P V t1 is co, amplify with Mik in many human cancers, yet. We also know that there are many mutations, and deletions, within this locus, in. Human cancer so this is a bit of a mystery, now because if you think about it if you're saying that this gene is very important for cancer growth then. Why would the cancer cell bother to make more copies of it and then, break it up us up as action, if it's actually good for the cancers growth so. I hope towards the end of this talk, some of these questions are become more obvious I, just. Want to remind you that Mick is a very important, aquitine it's, over expressed in nearly, 50%. Of human cancers it's, a key transcription, factor for cell growth and. We can mediate all the hallmarks, of cancer. Okay. So, one. Of the ways we realize, the, effect, of silencing, Pvt Wong with the guy RNA which is shown in the screen called. Sgrna. PBT, one isn't, an asset of competitive, cell growth if. You imagine that we have Sal star basically, controls, a wild-type and some, other cells or PV Jiwon silence, we mix them together we. Can imagine that the cell growth advantage will be manifested. I want, population, growing, at the expense of the other so. In this case we've labeled some, of these cells like the one silencing, PBT one with, a red color and the control cell silencing, irrelevant, genes. Was there with a green color if you make them together then. The red cells will grow out but, in another, control, some, of the truly relevant comparisons. The red and green cells we maintained their ratio over time neither, cell can grow better than the other, okay. So doing exactly, this experiment. We, found that willfully silence, Pvt, one in cancer cells showed, in red they, will basically grow, out and take over the population in, about seven days okay, but, in some control, of comparisons, basically, the red and green cells maintain their ratio so there's a potent, cell, growth advantage as rapidly, manifested.
That We can see in cell culture and this. Is also true in the animal. If, we inject these. Cancer, cells which have been tagged with a. Bioluminescence. Strategy, so this sounds basic will glow and report how many cells they are reject. Them into mice one. Side the, control, showing. Gray and the other side silencing. Pb2 aren't showing red you'll see that always, on the left the signal is brighter that. Just shows that in vivo that, there's a cell growth advantage for, these tumor cells so. Somehow removing, PB, to one transcription, has this, beneficial. Effect for the cancer cells. We. Wanted to know the molecular, mechanism, of this effect so we basically carried, out a, profiling. Method called RNA sequencing, looking, at all the differences, in RNA levels across, the entire transcript, oh and. What we found in this volcano. Shown here is that the, number one down regulator jean-louis, Islands PBT, one is Pvt, one as expected, that makes sense and the number one gene, does turn on it's, actually Mik this, gene that's 53, KB away, okay. This potent oncogene. So. We. Can further validate. Confirm, that result, by looking at protein, levels so, on the right hand side we're looking at loading controls, basically, everything looks the same and if, you silence, PBT 1 in column, bros 2 or 3 we see Mick protein, level goes up in concordance, with its, messenger RNA, so. It's good to remember the relationship between PBT, 1 promoter which we've been shutting down and Mick, as the, seesaw, relationship, he. Beat you on promoter goes down it. Goes up any, other experiments, when it goes down to promote TVC one promoter goes up. Okay. So so far so great we have this very interesting long non-coding RNA, locus we have this potent effect so. We were all ready to investigate, the RNA, and how does the RNA do his job but, then additional, experiments indicated. That the PBT one RNA, is somehow dispensable. Not necessary, for the effects that we're studying here I told. You about how we were using this, CRISPR, I to block, pvq, on transcription, there. Is a different, version of that technology, we, can use the dead cast 9 and use it as a row block downstream. Of the transcription of, the unit in this. Case you would not stop the, initiation, of transcription but. As the RNA polymerase is going along and we're running to the roadblock and then it would stop so your RNA would get truncated, okay, and that would also cost the RNA to go away on, the. Left hand side we're showing that in fact both, CRISPR, I Lord. This dead cast nitro block effectively. Reduces, the P V t1 RNA, levels, that we can measure basically they're absolutely. They're, nearly identical in, terms. Of getting rid of the r and p b t1 RNA, however. Only, stopping, transcription. From the p vq m promoter increased. Mik levels but, not the. But not giving that the RNA product of PBT want by this roadblock in, additional, experiments, looking at cell growth again, showed that only this former strategy, affecting. The promoter and hansel growth but not getting rid of the RNA. Well. We're, pretty stubborn so we kept going with, different methodologies, as. I showed you the, CRISPR, I method, blocking. Transcription. Increased. Mik increase al growth but not the roadblock to. Additional methods getting, rid of the PB tRNA, by antisense, oligonucleotides, or. Si RNA knockdown. Also. They, all effectively. Removed, the PBT one RNA, but did not affect, Mik transcription. In the same way so. We're forced to think about a different model for, how the pbq on locus could work as a, potential tumor suppressor. So. Here we turn to really the DNA, elements you, know if the PBT one locus and specifically. Thinking about how this regulatory, landscape would. Work on the DNA level in the three-dimensional, and genome context and we. Were aided by a technology. That my lab have recently developed called the enhancer connectome, this. Is a methodology, that allows us to a suppose, the enhancer, strength, and enhance, our targets, in the same experiment. What. We're doing is that we're taking cells, and. Fixing. Their, three-dimensional, contacts. Of DNA in the living cell, afterwards. We retreat, active, enhancers based. On a specific histone, modification. Called, histone h3 lysine, 27. Acetylene. When. We sequence these contact, maps we, can then create in a map of enhancer, promoter contacts, and this, method has some additional advantages. Shown. At the bottom here and I refer the audience to my. Talk part 1 in, epigenetic. Genomic technologies, where we go into more depth of other applications, for this technology.
In. This particular experiment, though we're focused on making PPT, 1 and so, the next image is so. Called a virtual 4c. Map, we're. Starting from a particular viewpoint in this case the. The NIC locus. We're interested, in anything any, active, enhancers that is touching the Nick locus and. What we discovered, is that this PBT. One gene, locus contains. Four, intragenic. Enhancers. These, are DNA, elements they'll, turn on other genes today, and they're shown here these. Four, arrows, here then, what we will further learn is that these four enhancers. Normally. Talk to the pv t1 promoter which. Is closer to these enhancers, but. If the p vq 1 promoter, is not active, these. Four enhancers, are still there they're still active and they reach over, and turn, on Mick instead okay, so that's summarized at the bottom these, four enhancers, normally activate, PT 1 and if, that P V 2 and promoters not firing the, enhancers, activate, Mick. So. This. Then really, additional. Experiments should really show that this isn't that how it's working if, we remove, the. Activity, of the PB t1 enhancers, then. The PB t 1 promoter silencing, no longer increases, Nick activity, no, longer drive cell growth if we, toggle Nick, then, P V t1 basically, gets more active and that again we believe depends, on the enhancers, and therefore. We're left with the idea of, an promoter. Competition. Model these, two nearby genes are both firing, the polymerase and goes to either the Nick or the P VG 1 promoter and these, four enhancers, normally, talk. To the PV T 1 promoter preferentially. But. If the p video and promoter is not, doing its job then. These. Enhancers. Would jump over to turn on Mick instead, so, the PV T 1 promoter, which is a specific DNA element, in the, linked RNA locus is really, like a fence right, it is a boundary, element, that, is inducible, and limits, the access of, the mcconkle. Gene to, these PB, t1 and tronic enhancers, so, this is a very novel concept because. Not. Only this is a boundary, element, not only is it offense there's, really a fence. Or a door, that you can control you can open and close as, I, will base on the amount of transcription, of this, link RNA locus. A very strong prediction, of this particular model is that Mick. And PBT one then, must, be working on the same piece of DNA right because we're talking about folding of the DNA on.
The Same allele so, we tested that with the following strategy, and. So. We. Used a cell. System. That's, derived, from two parents. Of very different genetic, backgrounds in, this, case a mouse f1. Hyper coming, from the 129, crossed, the caste, these are two different strains with lots and lots of DNA. Sequence differences, between them and therefore. In this offspring, has one chromosome, from 129, and one, chromosome from caste and. When we perform, either RNA, sequencing, or other kind of chromatin assays we can always tell which chromosome. Which allele, it is coming from and so in this case the 129, is always can be shown in pink, the caste in blue and, we can calculate a ratio which. Is called a D score you, can think about this discuss, a deviation score right, so if something is basically, exactly half. And half equal. 129. Caste is going to be 0 if, you see a big positive. Number is going, to be 1 all 129. Bias if, this is a big negative number it's, all cast okay. And. So we found is a very, interesting physiological, regulation. Of PBG one and Mik in. The, beginning, of development, for example in embryonic stem cells. We. Can map these, different activity so in every, row here is a different, profiling. Method and assembly. Showing two tracks with one color is the, 129, allele one color is to cast allele and then. This is looking at D chromatin, accessibility by, attack seek RNA. Sequencing, what's being transcribed and the histone marks k4, methylation, of promote active promoter mark and also, a canine, methylation, which is a silencing, mark so. In embryonic stem cells basically both, alleles, a pvt one are active, and basically. There's some basically. And the two of you also make up both firing, but at some, level okay. But. If these embryonic stem cells turn into, near a precursor cells in one cell. Clung all the daughter cells from the same allele we see something very interesting going on, now. The. Two alleles are no longer exactly, the same the. Blue allele here you see it's active, and the, red allele is not in terms of its promoter the other ileana, has a silencing, mark, so one ilio active, one allele silent, the, active allele of pvt one generates, lots of RNA, and the same allele, of Mick now has less RNA, compared, to its counterpart, that does not have those regulations, there's a one point four fold difference so, this really provides, the evidence, that, there's a mono allelic, regulation. And that pvt. One regulates, Mik on the same chromosome but not the other, okay. We, can look, across multiple, cell. Clones and look at how, when. How many copies of make being is, active, and. How many sorry, how many alleles, of PBT, one is active and how much Mik RNA is transcribed so. We can see cells with zero leop vt1, or one copy, Toledo's, active and there's, a corresponding, stepwise. Decrease. In Mik transcription, with more copies, of PBT. One being active, this. Is also shown in the following way if we, just count the allele, bias I told you about this deviation, D score okay, so basically we're. Calculating the, allele bias of pvt, one versus. The on the x axis versus, the leo bias of Mik on the y axis and, you see this inverse, relationship right. Whenever it more Mik is active, on that allele there's, less pvt one and conversely, more active pvt, one less. Make on the same allele so. I told you about the seesaw, analogy. And that, makes a lot of sense because, again, remember tea, to a promoter being active. Make us not and vice versa and these two. Players. Happy. On the same plank for them to play seesaw, and these, two elements have, been in the same chromosome for them to regulate each other insists. We. Also delve into the, raw of pv2 on promoter, in human, cancer genetics, so. Before. We, looked at the pvt one gene has a single, unit, but, really there's a new viewpoint, about the link RNA ask a series, of DNA elements suggest. I we should really think of individual, elements as the units in cancer genetics and. So we have the promoter and we have the enhancers, and we suggest, that enhancers. Was the oncogenic, driver the. Promoter is actually a tumor suppressor and, maybe what the cancer cells are actually breaking is just. The promoter and in fact we work with clinical colleagues to, examine, cancer. Genome sequencing, from breast cancers and, we discovered, that in fact the PV QM promoter is a hotspot just, a promoter is the hotspot of, the cancer mutations, and if you lie of all these different events together. Shown. On the right here you see that the commonality, is the PvP one promoter right this is the hotspot that's in common to all of them including deletions. Inversions, breakpoints, and so forth. We, went on to actually directly.
Prove, That. You, know P V t1 promoters, that a tumor suppressor and, won't have to find the minimal, unit. That's needed, and so. Some. Would show very talented postdoc carry, a Lashley genome editing at the PBG one locus, and then, we selected, four cells that would grow better and we discover that among. The cells that have a growth advantage now this. Included, a series of deletions. And the PB t1 promoter and we've discovered that a promoter, deletion. As small. As 6 bases. Is, enough, to, give a growth advantage and, increased, make transcription, okay, which was shown in the following slide. Here that we can document this, very potent effect on growth, and gene expression and, so. This the really defines kind, of a new Suri unit, in. Gene regulation and also you can search in that X. So. In summary what I've told you about is, the link RNA promoter as, a tumor, suppressor and, DNA element, it, the, discovery, came from using genome wide screens, and but, then doubling into the mechanism, we'd learn, about, the role of the 3d genome in shaping, the regulation, between the pbq, on enhancers, to Kevon tdq, on promoter and mick and also, this developmental, regulation, that. Is actually very interesting and, has and. Usual mono allelic. Aspect. The. Cancer, genetics, in what, we see in patients tell, us which elements, are really important, and driving, cancer progression and we live left with this model of this promoter, competition, the enhancers, being able to activate their PV q1 or Mick and vice versa and in, cancer what we're seeing is a systematic. Effort to. Basically steal, those enhancers, break, the PBG 1 promoter so make it have maximal, access to decent cancers and so this, is now raises new possibilities, of understanding, the system and targeting. Them and. To. Summarize this, part of my talk I. Want, to remind. Our viewers also, if the part 2 of the talk about linked RNA genes that act on the RNA level such, as exists, and so our, overall picture, now is that linked RNA genes may function both, as regulatory, RNAs, and as DNA elements and, specifically. Then linked RNA gene loss I can intimately, control chromatin, especially. Insists. On the same allele, at same chromosome, and finally. New. Technologies, enable, a systematic, interrogation of, linked, chromatin linked. RNA increment to interplay which is oftentimes very fruitful, understanding. Linked, biology, Linkara biology, and function. You.
2020-01-31 06:31


