Everything Matters | Gallium | Srabanti Chowdhury | Exploratorium
I. Would. Like to introduce shravanti. Chaudhary. She. Is a professor, of elected, by the way, this. Person here makes me realize how little I've accomplished, in my life she's, a professor of Electrical, and Computer Engineering at, the University of California, Davis and, as. A professor and technology. Enthusiast. She. Enjoys doing research, and teaching students, about new technologies, and how they impact our world as. And. As a mom of a five-year-old. She's. Getting good at juggling well, metaphorically, speaking can. You actually juggle. Ok. So. I'd like to have shravanti. Talker tell us about her work. Thank. You for that very nice introduction, and this is very exciting actually I should have asked all my students, to join but, probably. They're so bored of me I won't. Try that trick on them this time so, let's, continue. From where Iran has left and I believe even though we didn't quite discuss. Too much about our slides but you, know the. Way he actually sort. Of put together his gallium slides made, my job very easy, so. You will have to go, through the boring part right now how do we make gallium, nitride, something. That Ron already shared with us so I'll go straight into. A. Game, like you said there will be some overlap and, repetition, but that's, good that will make you remember this forever. So, gallium. Like Ron, has. Been talking about is the metal, the the, magic metal that makes that. You can actually melt in. Your hand and makes, the spoon disappear, just don't drink, the tea that was that, was basically Ron's plan we will we didn't go that far so that, it is quite non-toxic. So, that's good good news melting. Point reverb this, is a recap, so it's kind of mouth at. Less, than ninety degree, Fahrenheit, or less, than 30 degrees Celsius, in. The, sad news it doesn't, really exist in the elemental, form in the art so you don't find gallium, just like you can find silicon. To a great, degree like go to a near a beach and you find silicon, in silicates, here. In. A similar way kind of exists. With. The. Other, in, compounds, form like bauxite. Ores fare like that. Ron was talking about so, you can actually get gallium as a by-product, that doesn't make it that cheap but nonetheless it's a byproduct so, there's. A way to get gallium. Just. Kind of showing you. How we call it is it's a group. Tree. Okay, grow three element. And this. Guy's aluminum. Indium which fall in the same column, actually plays a very important, role in what, you just showed what we showed in semiconductors. Actually, okay so, I'll. Come, come, to that in a little bit so, let's do, a little bit of what. Gallium. Has produced, so far so, gallium in semiconductors. So I think, most of us are familiar about, semiconductors. Because almost, everything, that we touch. And feel. Today are either made, of are, made, of some form of semiconductor. Chip mostly, we known as silicon, but, there are other forms of semiconductors. And some. Of them as. It, is shown here. Do. Contain, gallium, okay so, gallium and arsenic, makes gallium, arsenide, gallium and, first, phosphorus. Makes gallium, phosphide, and then, gallium and nitrogen, gives, you gallium nitrite so these are called group five elements. And there's a group Group, three as I pointed out and you, can see how the, group 3 and group 5 that's a very nice ratio. Actually five plus three is, eight which is actually, what, makes the. A. Shell. Of a, compar, of an atom complete, that means five plus 3/8 is a closed. Shell structure. Okay close shell configuration. So that kind of says that these are stable, compounds, okay but. And, of course gallium arsenide has at, one point of time people thought gallium, arsenide, replace everything that we see in silicon. Or make with silicon that didn't happen, which. Is not. So good for gallium arsenide, enthusiasts. But silicon. People liked it that way. And, but. It exists, as Iran, was saying briefly, that, they do exist, in your cell phone so they use what is called a power amplifier in every handset you have a power amplifier the base stations, use, amplifiers. Those are made, of gallium arsenide. The. Problem. Is arsenic, is not such a friendly, element. So we are a little scared of arsenic, so, we thought that we might move.
All, Those applications to now, with, gallium, nitride. So things that were made of gallium arsenide, can, be made not everything, but a lot of this power amplifiers, are now made with gallium, nitride, and nitrogen, is much. More friendly and benign so it's. A good news so, some. Of the applications like I will, be mostly pointing to applications, are in near, infrared lasers. Solar. Cells some, very expensive. Solar, cells that are used in space, space missions, are made of gallium arsenide, high, speed radar, very. High frequency, anything above 250. Gigahertz need, gallium arsenide, because electrons, really zaps. Through the crystals, it's really, fast okay very very close to the speed of light which, is within electronics, means one, of the fastest, you can get and then, as I was saying cellphones in the base station, and handsets. Ok, so. Now. Let me talk a little more about the gallium nitrite this is something I work with we, make gallium nitride I'll show you some of the wafers that comes out of our lab. And. Just. In. Here it's basically, the opposite. Kind. Of the spectrum, I'm showing so now we are showing the 3 grew trees aluminum. Gallium and indium they, are actually called a3 nitride, family, because. They together make. Many, cool things happen, so aluminium bonds with nitrogen very strong bond by the way aluminium, nitride. Nitra and indium nitride and, that has quite changed, the world so the good news again they're non-toxic, and even, though, gallium. Doesn't exist in us. As ron, was alluding, to. Back. During his talk but there's a biocompatible, so, today, actually you can actually make a teeny-tiny, gallium. Nitride chip and you can, have, biocompatible. Applications. Things, are happening, like in coronal scoping and things like that you. Are actually making, a small tiny chip, of gallium, and you can digest it it's really, not going, to kill you I mean it probably will help you in the long run but so. So. Basically, this our this is very important, for us it's biocompatible. So a lot of the bio. Compatible. Applications, and. Medical. Applications are looking into this material okay okay. So. Let me talk a little bit about the gallium nitrate and the led so, I actually studied, at UC Santa Barbara where, LED, was invented. Shuji Nakamura, whose, picture, was, shared. In the previous, part of this talk. Happens. To be one of my professor, and actually, I thank. Him for giving sharing some of his slides here so I'm going to show some of his slides, and. I. Cannot. Say. Enough about the LED but. What, can but, it as we all know it has quite changed, and transformed, the world we see today so. Light. Lumens. Per watt one, lumen is basically, the. Amount of light that a source emits, is, expressed. In lumens, okay. And. As you can see how the. Tragic trajectory, went from an oil lamp to, an incandescent bulb, to, a fluorescent, lamp and now we are in this led, era. With, over. 300. Lumens per watt, okay. This, is a very, very steep rise a, 60. Watt, incandescent. LED. Bulb produces almost. 860. Lumens, okay which, if you make it with an LED to I will. Consume. Less than 5 watts so that kind of tells you how efficient, lighting, is because, of LED. Now how that how did that happen, so. Again. This is a little bit of a recap the, white LED was possible, because, of gallium, nitride so, this is a white LED a, packaged. LED and this is the bulk Ron has displayed, it thanks, Ron for that, helps us so. The gain led today is the basis of more, than 15 billion and growing as you can as you can, understand. That population. Increases, and, places. Where you cannot, get the gridlines, or the electricity cannot, be transferred, to grid you, can still get hold of a battery, and you can light up an LED right so this has a. Huge. Significance. In today's world ok, now a very, quick cartoon, of, what. And how an LED works I have. To share this I mean this is something. My, undergrad, students, who would not appreciate very, much, probably, anymore but basically. What. It what this shows is it's a battery. You you. Bias. Or you, put voltage across this material. And this is what the red part is what is called the active part of the LED ok you, have let's, call this a substrate. Which is basically the foundation by the way gallium nitride, is grown on a substrate, ok so this, is the foundation, of the whole chip, then.
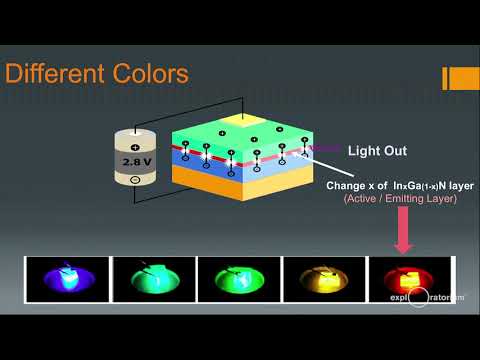
You Grow, the, source of electrons, or also called an n-type, material, or n-type, layer, you. Use, sandwich, the active region ok the active or the emitting light emitting, layer and then, you have a p-type, region okay. So. Both. P and n can be made out of gallium, nitride and this. Active, region actually is made up of gallium, nitride, but you put a little bit of India minute and that's the trick ok I'll show you how so, basically. That. Emits, the light what, happens is this electrons, and holes the electrons are sourced. By the end layer the, holes of source by the P layer they combine, they, recombine we, call it and they, give out the light okay and. Going. To, a more. Fab. Like picture, so this is an actual chip, okay, I have some of the chips I'll demonstrate. Here, and this is the packaged, blue light okay, so this is from the chip to a package, and. Well. How you get the different colors is even more interesting so, what do you get what you do is you you. Change that remember, what I said here, in the active region you. Have indium. Okay, so you change, the, composition. Of indium, into. The gallium nitrate and by that you can change the colors so you can you can add more indium, and it becomes red, well if we're not yet here a lot of research is going in this region by the way if you, just have a pure, gallium nitride, you get sort. Of violet. Okay close, to UV, kind of light then, you add a little more indium you get more, sort. Of a Sian, then. You get a true green then. You go to yellow, you keep adding indium. More and more to be a liam Nitra, and you get towards. Red okay and believe. It or not a lot of work, is happening here it's one of the, one. Of the hotbed, of research, right now how do we make a red LED for, all from gallium nitride. Okay why, because. Ideally we want to have RGB. The three primary colors if you have three primary colors you can mix them in any proportions, and you can get any kind of light out of it right so that is why we want if, so that we can integrate everything we, want in one, chip we, want all our. G&B. All, in, gallium nitride okay where. Are we today we, are here we, have nicely, achieved this actually, so we have blue and blue, light and yellow. Light and we. Can mix them to produce white light and this is exactly how white light was produced at Bob you see that, has blue and. Yellow. To. Make it, white. Okay. So this is basically what what, it does you, have a blue LED which gives you a blue light you you. Convert. The blue into yellow, using, phosphorus. And you then mix the two to, form white. This can be done with other colors also okay and, this is how the white LED was, producing I'm going to show you something, that we get.
From The. Lab. And. This. Is one of this is only one, chip. Of LED, okay and I'm just taking a battery. Let. Me make sure so. I have, a positive and a negative, and. I. Don't want to. That's. Bright so this is one chip. Okay. Accompany, very generously. Packaged, it for us and I was lucky that this happened, right before the stock was scheduled so I was able to just get, it from my lab so. There, you see it's one LED. Not. How you it. Blinds me almost and, so, this is pure blue correct, now, we add the phosphor, when. We add the phosphor. This. Is a warm white, so. This. Is one, big, LED. Now. You make. You. Guys can see right so, that. Is what you see in those bolts so these are I, hope. Some cool examples how. And then, you can of course make cool. White this is also done. Using. Several, of those chips okay. And this. Is more you, can see the, cool and the warm just indicates the color temperature this, is a cool white that was the warmest way but. This is as. Close. To, a. Wafer. Stage you can see basically, of. Gallium nitrate as close to the you. Know fundament. Or basics. Also. Visa. Okay. So. Moving forward, like, me. So. So. I hope that is. That, is exactly how we are getting white LED, and I hope this kind of shed, some light how, we see today's all the, white bulbs that you can buy in a Home Depot. So. Of. Course very well deserve the, three people. Who are the three guys who were responsible, for inventing, the blue. LED and. Were. Awarded, the Nobel Prize in 2014. And this is very important, one of the reason I kind of. Highlighted. This that, for inventing, invention. Of efficient, blue light-emitting, diodes. Which has enabled, bright, and energy-saving. White. Light sources, so it wasn't, because they invented LED because LED, was invented, way, before. The. Gallium, nitride LED was invented, but it's because this. Enabled, white light and once you have white light it, completely, changes. The world perhaps. Only. I, mean. The perhaps only explain credited. By a Nobel. Prize so. This. Is how the trajectory. Goes, the. Rise. Of this LED market as you can see by again has. Basically. Has. A very, very sharp and steep price and. I mentioned, I repeat it is basis, of a fifteen billion dollar plus market. And growing so. Moving. Forward there, are other emerging. Applications that, are, coming these are all part of the research some of it which we do in university, some of it is done in the industry but, other than decorative, lighting automobile. Red lighting displays. There. Are agriculture. People have shown that some. Of the herbs. At, plants, grow much better at a certain, wavelength of light, indoor. Lighting and then there is another thing which is very coming. To. Practice, a lot is basically LED, visible, light communication, or. VLC, so. The next generation of lighting, is actually, starts with a blue laser and some, of the automobile, headlight, is right now with a blue. Laser that, has already started, okay. So final. Part of my talk, this is something that I work, for in, my lab. This, is about power conversion, I won't, go too much, into the details, but let me explain the way this things work you take, from one stay and you, convert, into another, state and you do it by basically chopping.
The Energy, in. And then, re using. Water called switches, and then, reassembling. Them into. The desired, form at the output okay that's that's, what is called. A power conversion, so you can take an DC, convert. Into an AC or, you can take. 150. Volts convert, it into a 12 volts okay so these are called power conversion, and what. Makes par conversion, possible are this tiny little things called switches okay so, I make, switches in, my lab my students, make switches, in lab and how do we make them we, make it a game with the, magic, material called. Gallium, nitrite, which, is, one. Of the. Most. Promising, material that that. Can. Change. The, way we see power conversion today so just, so that you know the. Amount of energy we lose in. Efficient. Electronic. Switches, is more. Than what we produced by hydro, and. Wind and. Solar together so this is the loss and this. Loss is, more than what we generate, by renewables, so, that is because. We are using silicon. Electronics. Silicon electrics, has brought us here which, is great but. We have a lot more to do and one. Of the thing that we do with. What, we're trying to do is make things more efficient, just. Like in the lighting it happened we are trying to do that in power conversion so, we use gallium, nitride, for, switches, this, is a fantastic, testbed. Forget showing gallium gallium nitride. Are basically. Showing. How power conversion works there are many, components, but one component you can understand. Easily is on board, chargers, there's, many other many other, especially. In. In the world, of or or in, the generation, of autonomous, driving. You. Will there, are much. More applications. That hasn't happened, with silicon and will never happen with silicon their gallium, nitrate can play a very big role a wireless, charging, is one of them okay, I'll. Go. To. This, smaller, slide, which, has a lot of data but all I would ask you to kind. Of pay attention is the, reason, why gallium, nitride, is helping, in Seoul, and this, host, of application, is by, running. Thing at higher, frequency. Because, when you can make things go fast, you, reduce, size okay when you reduce size you, make, things portable, you make things compact, and I think everyone. In this room will appreciate what, it means to. Reduce. Form, factor what it is to reduce size so. That. Is one one, motivation. To do gallium, nitride the other motivation, is you can make things run, hotter so all, this electronics. When you convert power, from one state. To another state produces. The, waste, you know what the wasted that's that is the heat so if you touch the charger, of your laptop after, two hours of working you will feel the, heat and that's the wasted energy what. We are trying to do is we are trying to reduce that waste by, going, to a more efficient. Semiconductors. Okay so there. Are certain, things that are happening today, like photovoltaic, inverter, this, is a this is the size by silicon, it becomes, less, than half it reduces, cost ultimately, so. It's. A very important, saves, energy as I, said that, power conversion, actually. Wastes, energy more, than produced, by renewables. That's a very alarming, factor. Moving. To. The very very last segment, of my talk the, lab so, I want to show you a few things this. Is how we grow gallium nitride. The chemical, reaction is we take ammonia, we, split it that are called cracking, ammonia. Cracks around 600. Degrees C and we, use gallium is a tri, methyl gallium T, ma it's called so it's so basically that metal organic. Compound. We, crack ammonia, we, take the nitrogen. And then, we form gallium nitride. And other, product, is met him. This. Is how the reactor. Looks does so this is actually from my lab. This. Is where we are making gallium nitride, and this, is the product so it's kind of the, you. Know how. It. Looks when, it comes out the reactor um these. Are called MOC medium metal organic chemical vapor, deposition reactor. And we, Grogan, unlike. You. Know many. Other semiconductors. Where you, can actually. Have. You. Can actually extract out, those. Compounds. Gallium nitride needs to be synthesized, chemically. And that is why we call it we grow we grow gallium, nitride, and this, from from gang wafer, to.
Transistors. And I see a lot. Of steps happen between here and there, and one, important, thing I want to bring up the, reason gallium nitrate became so popular in power conversion is because, if we were able to grow it on silicon, silicon. Is dirt cheap and that is extremely, important, because with, every, technology we want to reduce size, and make it cheaper so how we will get there the only way we can do it is if we take help of silicon, so we have started growing gallium nitride and silicon, which have very much can be very big wafer so silicon, can be you know ten inch wafers, right, in diameters so, that's. This is a gallium, nitrate on silicon, there are other substrates. You can grow gallium nitride, sapphire, I have an example here where, we basically grow gallium. Nitride, on sapphire. Okay. And this. Is basically how it looks when it comes out of the reactor and this is not just gallium. Nitride it is grown on a, foreign, material as, we, call it or a substrate, called remember, that foundation, if you remember like, in, one of my slides I call it a foundation, that foundation, is very critical and that is called a substrate this, this, is the how a transistor, look, my students. Work very hard to make them very. And, then, we can make icees. Basically. We can make circuits, so, on and so forth the, very important, factor smaller form factor higher operating, temperature. Lowering. Cost lower, in energy what else do you want, so, basically. So. So. That. Concludes my I. Don't. Know, music but that's okay so. This is the vision of tomorrow okay this is how crazy. Things, will look like, so. No nothing is hitting even though cops are happy because they don't have to, do any they. Don't need a job actually at that point from they shouldn't say that, well, whatever. So. This, is how crazy the world is going to get and, yet we will all live right, so, why what do we need we need very. Smart technology, which cannot happen without gallium, nitride so. All, this Leiter's. Radar, power converter sensors, detectives, wireless, charging they are and and this, is so, true actually they are all probably. 80%. Of them will. Still be silicon but, 20-person is a lot of all, that okay, and for, that 100, percent you need gallium, so, with with. That I would just hang call my group this is my research group and, I would definitely, acknowledge for. People who helped, me gather, some of the slides. And. I would of course a lot of this research was possible. By the government, sponsored. Government. Agencies who find this kind of research so, I'll conclude that and thank you I'm happy to have take any questions.
2018-07-25 06:14



Amazing video, as always.