Kavli Institute for Brain and Mind: Imaging the Brain - Lippincott-Schwartz
So. I'm gonna be talking about the. Complexity. Of the. Single, cells that, make up the brain we've, heard a lot about. Neuronal. Populations. And how they're organized, which is quite. Amazing but, if you go if you zoom in and look, at any one of those cells, whether they're astrocytes. Or neurons or, microglia. Within, the. Brain inside those. Cells is a. Plethora of. Sub. Cellular organelles. That. Are. Playing. A, huge, role in how the brain is operating, I. Just want to draw your attention to some of these organelles, who's. Who. If they don't function properly can, lead to many of the neurodegenerative, diseases that we're aware of so for instance mitochondria. That you can see here. Defects. In mitochondria, lead to Parkinson's many, of the Parkinson's, disorders. Lysosomes. Proteins. That comprise some of these lysosomes, are, responsible. For frontal temporal disorders. And the, endoplasmic reticulum, which which, you will hear a lot about today is. Very. Much at play in. Where. Mutations. In proteins that, shape that organelle. Underlies. A variety, of spastic, paraplegia. Disorders. So in, order for us to really get I think at this neurodegenerative. Aspect. Of the. Brain and how it ages, and deteriorates. We, need to understand, how these organelles, that comprise. The, cellular, components. Which is the unit of, that. The whole system of the brain are. Organized. So. This. Is a classic, transmission, electron. Micrograph a 90 nanometer slice. Through. A cell to, really. Reinforce the. Complexity. Of these organelles. What. We want to know is. More. Information about the. Three-dimensional, organization. Of these. Organelles. And how, proteins, are dynamically, distributed. That's. One thing we want to know and I'm going to be talking, about other, layers, of information, that. We, are now beginning to get in terms of the organization. Of this, system. Thanks. To high end microscopy. Technology. So. Let's start with the 3d organelle, shapes. This. Is as I show as I mentioned, is a transmission, electron, micrograph. Slice, of 90 nanometer 's what, that means is within that 90 nanometer. Slab. You, have no Z information. Everything's, flattened, but. Thanks to, technologies. Like the focused, ion beam scanning, knowing. System. That in combination. With scanning. Electron microscopy we. Can now slice, through, the cell at very thin. Sections. For a nanometer in this case and, we've. Used that on collaboration. With Harold's beautiful. Fib systems. At, Genelia to, begin. Slicing through, parts, of the cell to, reconstruct. Particular, organelles, in order to understand, how these organelles, are shaped in 3d and how, they communicate with, other organelles, what. You're looking at here is just the fine complex. Architecture. Of the endoplasmic reticulum, in a, small the. Edge of the cell in, this, panel right here. What. You're looking at is the confocal, volume, that we've sliced. In. What, you would see if you were using a confocal microscope. So you can see there's a huge difference between the. Reality of this organelle, as revealed. By imaging, at this very fine, three-dimensional. Architecture. Versus. The. Typical, image that you would get if you're using conventional. Confocal. Microscopes. Now. We've been working. With perrault. To look, in more detail at these. Organelles, in. This case we're. Looking at plasma, membrane mitochondria. ER, and the, songs that we, can segment out as. You mill through the, slices, this. Is a two micron, volume of the cell, four. Nanometer, voxels, and. You, can color code each of these different organelles, to, see how they're, arranged, relative, to each other now. The, mitochondria. That you can see here in green are, intimately. Communicating. With the ER, which. Is shown in red and there's, a lot of crosstalk between these, two organelles, that's, absolutely, critical for calcium handling, in the cell for. Reactive. Oxygen. Exchange. As well, as lipid, and other types of communication well. How do we localize, proteins, on. These organelles, and the. Approach that we've been taking, really. Builds from really. The twenty years or more of work, that people done using fluorescent. Protein, technology. Which, allows you to tag proteins, of interest and then, look at how they're distributed, now. In order for us to get high resolution. Protein. As well as lipid, distribution. To. Understand. The fine architecture. In these. In. A three dimensional section. Of a cell we've. Applied, lattice, light sheet microscopy developed. By Eric Mexican.
Colleagues Where. Essentially, you, take ultra. Thin vessel, beams to. Create a thin, 2d, optical, lattice which. Is then used as a sheet, to pass through, your. Cell, of interest. What, this allows is, ultra. Thin slicing. Through a cell, which. Is at a much smaller scale than, big. Neuronal, slices. Sections. And. Because. We're we have such a thin light sheet that's, basically, almost the same dimension. As the XY, lateral, resolution that, you get with the microscope, you have isotropic, resolution. And again, it's at low it's relatively, low to photo toxicity. Because it's. A lattice sheet, of light rather than a full, sheet so. We combined, the. Lattice light sheet microscope, with. A point. Localization. Or. Like imaging. Where, individual. Molecules, in this case we're looking at lipid, molecules, that, bind and then dissociate. From. Membranes. And whenever they're bound they. Create. A spot, on the surface of that membrane that, we can fit with. Very high accuracy, by, point. PSF. Centroid. Fitting, and when, we do that we can reconstruct, the. Entire sort. Of organelle. Distribution. Map it out in 3d through. Lattice, light sheet imaging, with. A combination of. Plotting. Out all the individual, distributions of these, lipids, that have been using. Super-resolution. Imaging, docked in and and. Put, in place now. Now, that we have an image, of all these organelles, and. This you can see mitochondria. Here, and this, web-like, structure. Represents, anaplasma, reticulum, we. Can now come in and dock in particular. Proteins, of interest I'm. Just going to zoom in on this area here because what, we were particularly interested. In or were interested in in this study was, how, Enda. Proteins. That are part of the endoplasmic reticulum. Which. Is this. Large structure. That expands, throughout the cytoplasm as this tubular. Meshwork. How, its organized, and so, what we can do, with. This technology is. Superimpose. Our. Distributions. Of fluorescent, proteins, that we've specifically. Genetically. Engineered. And tagged onto. Our a. Fluorescently. Acquired, image using. This lattice light sheet system, to. Dock. In where these proteins are localized, and this, is an example where we, have again. All of the membranes of this of the cell that have been painted, out if you will using. The, lipid our lipid probe and single molecule super resolution imaging, and we're, now correlating. It with a diffraction-limited, image of. Sex, 61 beta tagged. With an M emerald, fluorescent, probe and, you can see how, they align, this. Is exciting, to us because it, really sets the stage for beginning. To, investigate, a whole, slew, of different proteins, and how they localize, within. This cellular, system. So. We. Think that fits in and lattice light cheap paint or palm-like. Correlative. Approaches. Will. Allow us to really, gain deeper information, about, how all of these organelles, are shaped. In three, dimensions, and how different proteins might distribute, on them but. One of the challenges, that we still face is, how. Many. Organelles, are arranged. Relative, to each other in a, living. Cell context. In. Particular, that's important, for understanding how. Different. Organelles, are, contacting. Each other and communicating. With each other we. Know for instance the, endoplasmic, reticulum, which is rep. This. Sort of snake-like, structure. That you can see in this en image is, contacting. Virtually. Every other organelle. Within the cell and. Communicating. With those organelles, through lipid, trafficking, Ross. Essentially. Exchange. Of reactive oxygen species, calcium. Signaling, many. Other types of communication, is going on between these organelles, and our, problem, with trying to understand, that is that, we haven't been able to look samel, taneous Li at all. Of these organelles, in a, living cell, context. We can see them clearly. With electron microscopy but. If we do fluorescent. Time-lapse imaging, we're, limited, to imaging. Two, or three of these of. These organelles, at one, time because. Of, this, problem of overlap. In emission, spectra, in, among. The different floor fluorescent, proteins that are available so. Here we have classic. Fluorescent. Proteins. That. Have, different, emission spectrum, cfp, GFP yfp, these. Are the fish, spectra, for each of these different fluorescent, probes and the. Problem, is their. Emission, spectrum, is overlapping. And what, that means is, that if, you tag a. Particular. Organelle, with. Different, fluorescent probes, like, the ones that I just mentioned which are the most widely used fluorescent.
Probes And then. You, image. Essentially. You go across the emission, spectrum, to, look at that at, any particular wavelength, of light which. Population. Of organelle. You've you've. Looked. At you can see that, you have overlap. At any particular wavelength. So that means if, you were imaging, for, instance at 4:38. You'd see three, different from. You you would not be able to distinguish mitochondria, or and lysosomes, from each other it would just be one big blur. And hence. You could not distinguish, how these different organelles are behaving, relative, to each other, so. To overcome that challenge two. Postdocs in the lab, decided. To employ a technology. Called multi spectral imaging, to, try to unravel, this. Overlapping. Emission, spectrum, and the. Strategy. That they used was essentially. Take. If you know the the emission, spectra of each of these different fluorophores you. Can then query, an observed, pixel, spectrum, that's a combination, of one. Or more of these fluorophores, and then use linear. Unmixing, to, decipher. What combination. Of and. In what abundance. Any. One. Or two, or more of these floors, would give rise to this particular, spectrum and. Using. That we can at each pixel. Of our image, unmix. To, determine, which floor four is giving, rise to the signal that we're observing at, that pixel, and so, we've combined that with lattice, light sheet microscopy to. Be able to image. Simultaneously. Six. Different organelles. Within, the cell over time, in, order to do that we've essentially introduced, six different laser lines at up that cover this the visible, spectrum, that allows us to excite, the, fluorophore that, we've tagged on each one of these different organelles. And then, we do our linear on mixing algorithms. To, determine. Each. The. Specific. Spectra, associated. With. Each, organelle. And this. Is what you can see in the case of these six different organelles that we've, introduced. Fluorescent, tags, for now. This is in a single cell so we can superimpose all, of, these signals, on top of each other to samel. Taneous li c, in a three-dimensional. Space because, we're using that lattice light sheet to, move through the whole volume of the cell how, all of these organelles, are distributed. Now. With this technique. We can begin to. Zone. In on. Really. Specific, measurements. In terms of organelle. Distribution. Localization. Essentially. Connectivity. This. Is just a, set. Of values, for the. Number of organelles, that each of these different. Populations. Of organelles represent so for instance on average. In the cell that we're looking at there's about 89. Lysosomes. 186. Peroxisomes. 157. Lipid, droplets. The. ER, occupies, by far the largest volume in this cell, among all of these different organelles it's about 30. Times the size of the Golgi apparatus which. Is involved, in the, secretory pathway, eight. To nine times the size of the, of the mitochondria, which is involved, in energy, production, within, the cell now. We. Can also come in and segment, these, individual, organelles to, look. At how they, are. Connected. To each other how they're contacting, each other in order, to begin, to understand. The, communication, the cross communication, or cross talk in, activity, that we know is so important, for how cells. Are operating and. Communicating. With other cells in their environment, for. Instance the endoplasmic reticulum. Controls. The secretory pathway, together. With the goal chap. It's what's secreting. The, parent the the paranormal, network that we, heard earlier about so under, it's critical, for us to really understand. How these organelles, behave. Relative, to each other, and from. These types of segments and images we, can create what we've, been able to describe. What we call the organelle interactome. Where, we, just measure the pairwise contacts. Between these, different organelles, in. In. Our images. Of these cells and from. That we can see the, frequency of, communication. That. Different organelles have, with, each other you, can see here that the ER is by far the most communicative. Of all of the organelles it's contacting. Everyone. Importantly. If you look at a single cell over time, you. Can see that this organelle, interactome, is conserved, over a fairly significant. Periods of time and that is. Despite. The fact that any, particular contact. That we see is relatively. Transient. I should, emphasize that this interactome. Changes. Dramatically, if we, perturb, the cell in in different ways we can deeply mirai's microtubules, or starve. Cells in different ways and we dramatically, change. The way these organelles, are interacting. With each other, now. This is a movie where we've segmented, out the mitochondria. As. An example, of a. An. Organelle, that is intimately. Communicating. With, the endoplasmic reticulum.
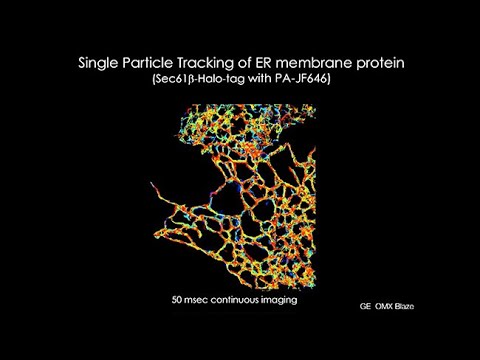
On The, right hand side represents. The. Surface, all. Of the surface, sites of mitochondria. Where, we see ER signal. So. The ER is wrapping, around the. Surface of the mitochondria. And, intimately, communicating. With virtually all of those, mitochondrial. Elements, that we see in this cell. This. Contact, we think is what's, up, we. Think there's calcium flux between the ER and mitochondria. That calcium, is playing, an important. Role for mitochondrial. Output, how much energy, is, being produced by the mitochondria we. Also know there's lipid, and cholesterol being transferred. Across, these contact. Sites and importantly. Reactive. Oxygen. Reactive. Oxygen species as being trafficked. Across those contacts, which. Could, play a big role in the. Disulfide. Bond formation and, protein. Remodeling. Occurring, in the endoplasmic reticulum, now. Let's focus in on the ER the ER for, a second it, occupies. 25%. Of this cell cytoplasm. We, can measure that using our light lattice light sheets reconstruct, three-dimensional reconstruction. What. Is interesting is that if we look. At we plot out the position, of the ER over, a 15-minute. Time period, which you can see in this movie what. We find is that the ER has pretty much explored, the, entire. Cytoplasm. Over, just 15, minutes so, it is a very, dynamic. Organelle. That has, lots. Of, capability. Of communicating, now. In my final 2, minutes or 1 minute I've. Got a I want to take you through how, fast these organelles, can. Move. We, know that. They. Can the the ER has the capability, of exploring, that cytoplasm. Let's look at its dynamics, at higher, resolution. We, can do this using a turf sim system, we, can see these, tubular. Matrices. Move, incredibly, fast, interestingly. The. Tubes themselves undergo. An oscillatory, activity. That's. ATP, and gtp, dependent. So it's not just thermally, driven, and. Finally. We can actually come in and start. Mapping. Out, individual. Proteins, that. Move, or diffuse, along, the surface of the endoplasmic reticulum, that's what you're seeing here each of these yellow spots, represent. A halo tagged protein. That, is associated, with the membrane of the ER and we can begin to map, out the trajectories. Of the proteins and that's shown here for. Another ER resident, protein sex 61, beta. If, you sum up all of these trajectories you. See that these proteins, and this is a transmembrane, protein, you're looking at will, explore, the whole, surface. Of that ER freely, now. In, my final. Movie. Here I just want to show here's an example where, we're. Mapping out a. Protein. That actually, interacts, it's, on the surface of the ER but it is part. Of a tethering, complex, that, brings, the ER close to the mitochondria, and what, we can see is when, we track these, individual. Molecules, as they diffuse, across, the, surface of the ER we, can see that as they move across the area of the ER that's in close proximity of mitochondria. They they, slow down significantly. Consistent, with a transient, interaction. Of, of. This tethering, protein with, the target, protein on. Mitochondria. So. With that I want to end and say that this, field is really, being. Significantly. Impacted. By, the high, resolution technology. That's now available, that. Scans, from you know electron, microscopy up, to the fast. Imaging.
Technology. People. In my lab have been greatly impacted, by the. Physicists. At Genelia Eric Betts ik and Harold in particular, who, really provided, the technology. That allows us to do this type of work thank, you.
2018-02-18 16:00


